3. Predict the major products for the following reactions. Pay careful attention when orientation is a...
Fantastic news! We've Found the answer you've been seeking!
Question:
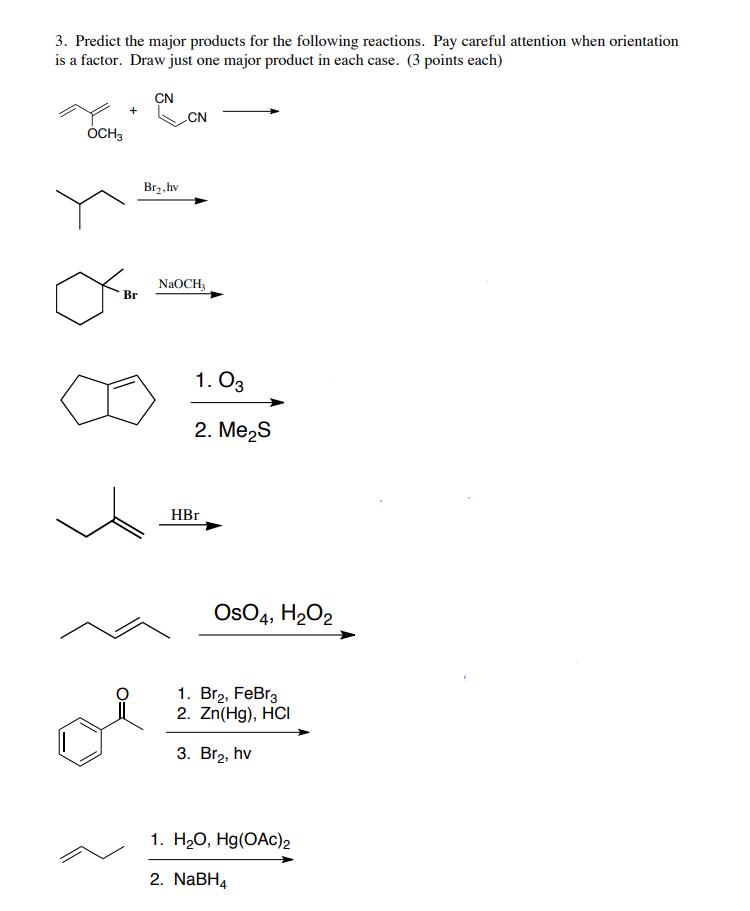
Transcribed Image Text:
3. Predict the major products for the following reactions. Pay careful attention when orientation is a factor. Draw just one major product in each case. (3 points each) OCH3 Br CN Br₂, hv CN NaOCH₂ 1.03 2. Me₂S HBr OSO4, H₂O2 1. Br₂, FeBr3 2. Zn(Hg), HCI 3. Br₂, hv 1. H₂O, Hg(OAC)2 2. NaBH4 3. Predict the major products for the following reactions. Pay careful attention when orientation is a factor. Draw just one major product in each case. (3 points each) OCH3 Br CN Br₂, hv CN NaOCH₂ 1.03 2. Me₂S HBr OSO4, H₂O2 1. Br₂, FeBr3 2. Zn(Hg), HCI 3. Br₂, hv 1. H₂O, Hg(OAC)2 2. NaBH4
Expert Answer:
Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
Predict the major products formed when: (a) Toluene is sulfonated. (b) Benzoic acid is nitrated. (c) Nitrobenzene is brominated. (d) Isopropylbenzene reacts with acetyl chloride and AlCl3. If the...
-
Predict the major products of dehydration catalyzed by sulfuric acid. (a) Hexan-1-ol (b) Hexan-2-ol (c) Pentan-3-ol (d) 1-methylcyclopentanol (e) Cyclopentylmethanol (f) 2-methylcyclopentanol
-
Predict the major products of the following reactions: (a) (c) (e) Product of (c) + H2O Product of (c) + H2 (1 molar equivalent) (g) Product of (f) HCI Styrene EtONa HA, heat OH peroxides HA heat Pt...
-
Barnaby is a farmer who receives two-thirds of his total income from the sale of oranges. After two consecutive years of early frosts that destroyed his orange harvest, Barnaby found himself deep in...
-
In the world of trendsetting fashion, instinct and marketing savvy are prerequisites to success. Jordan Ellis had both. During 2012, his international casual-wear company, Encore, rocketed to $300...
-
The radioactive decay of a substance is modelled by the formula R = 140e kt t 0 where R is a measure of radioactivity (in counts per minute) at time t days, and k is a constant. a. Explain briefly...
-
A social venture is a business that seeks to solve a social problem and provide a social good. A social good is a product or service that benefits society. Clean air, clean water, literacy, and...
-
Cairns owns 75 percent of the voting stock of Hamilton, Inc. The parents interest was acquired several years ago on the date that the subsidiary was formed. Consequently, no goodwill or other...
-
29. If A 21+j+k and B=1+2j+2k, find the magnitude of component of (A+B) along B: (1) 4 unit (2) 5 unit (3) 6 unit (4) 7 unit 30. Which of the following group of forces cannot produce the resultant of...
-
Kayak Company budgeted the following cash receipts (excluding cash receipts from loans received) and cash payments (excluding cash payments for loan principal and interest payments) for the first...
-
The accounts receivable of DEF Corporation were stated at P1,967,000 in its balance sheet as of December 31, 2022. Upon analysis, the unadjusted balance of the account was found to consist of the...
-
Explain how theory can contribute to explaining collaboration between firms in general and how theory can support supply chain practitioners in performing their job.
-
Describe how the supply chain concept has extended the traditional view on logistics.
-
How has consumer behavior, especially in the western industrialized nations, been driven by new logistical developments and what challenges have emerged in recent years?
-
Your company has recently moved from single sourcing to dual sourcing, adding a second supplier for a key component of your product. While the relation with the incumbent supplier is based on a long...
-
Discuss the fundamental traits of neoclassical theory and briefly explain why it cannot explain the existence of supply chains.
-
Auditing is a prestigious profession in modern society and therefore they are expected to provide a high quality service to the stakeholders. Audit quality is highly regulated nationally and...
-
suppose a nickel-contaminated soil 15 cm deep contained 800 mg/kg Ni, Vegetation was planted to remove the nickel by phytoremediation. The above-ground plant parts average 1% Ni on a dry-weight bas...
-
Classify the following amines as primary, secondary, or tertiary: (a) (b) (c) (d) (e) (f) H IN NH2 HN
-
Write the following as net ionic equations and designate the nucleophile, substrate, and leaving group in each reaction: (a) CH3I + CH3CH2ONa CH3OCH2CH3 + NaI (b) NaI + CH3CH2Br CH3CH2I + NaBr (c) 2...
-
Draw a three-dimensional orbital representation for each of the following molecules, indicate whether each bond in it is a s or p bond, and provide the hybridization for each non-hydrogen atom. (a)...
-
What is meant by corporate governance and why is it important?
-
Continuing to focus on evidence associated with the act, concealment, and conversion, use the evidentiary material to continue the examination. In addition, the examiner also starts to think of terms...
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. Determine the ERR for this project. b. Is this project economically attractive? EOY 0 2 3 4 5 6 NCF -$100 $15 $15 $15...

Study smarter with the SolutionInn App



