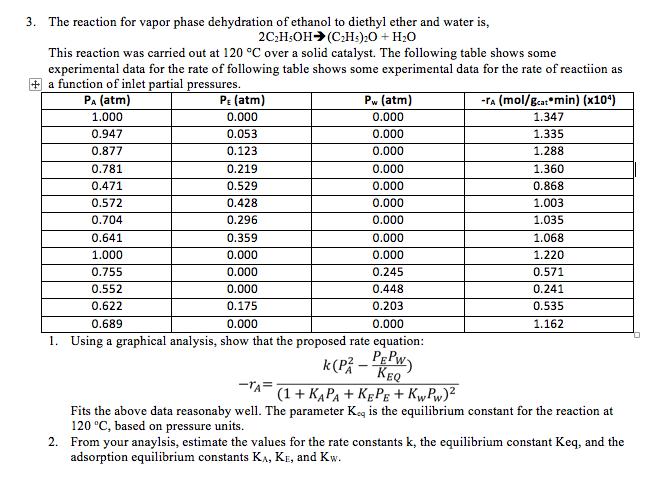
3. The reaction for vapor phase dehydration of ethanol to diethyl ether and water is, 2C;H:OH...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
3. The reaction for vapor phase dehydration of ethanol to diethyl ether and water is, 2C;H:OH (C:Hs):0 + H20 This reaction was carried out at 120 °C over a solid catalyst. The following table shows some experimental data for the rate of following table shows some experimental data for the rate of reactiion as + a function of inlet partial pressures. Pa (atm) PE (atm) Pw (atm) -TA (mol/g.a: min) (x10) 1.000 0.000 0.000 1.347 0.947 0.053 0.000 1.335 0.877 0.123 0.000 1.288 0.781 0.219 0.000 1.360 0.471 0.529 0.000 0.868 0.572 0.428 0.000 1.003 0.704 0.296 0.000 1.035 0.641 0.359 0.000 1.068 1.000 0.000 0.000 1.220 0.755 0.000 0.245 0.571 0.552 0.000 0.448 0.241 0.622 0.175 0.203 0.535 0.689 0.000 0.000 1.162 1. Using a graphical analysis, show that the proposed rate equation: k(P - Pw KEQ -TA= (1+ KAPA + KĘPE + Kw, Pw)2 Fits the above data reasonaby well. The parameter Keg is the equilibrium constant for the reaction at 120 °C, based on pressure units. 2. From your anaylsis, estimate the values for the rate constants k, the equilibrium constant Keq, and the adsorption equilibrium constants KA, KE, and Kw. 3. The reaction for vapor phase dehydration of ethanol to diethyl ether and water is, 2C;H:OH (C:Hs):0 + H20 This reaction was carried out at 120 °C over a solid catalyst. The following table shows some experimental data for the rate of following table shows some experimental data for the rate of reactiion as + a function of inlet partial pressures. Pa (atm) PE (atm) Pw (atm) -TA (mol/g.a: min) (x10) 1.000 0.000 0.000 1.347 0.947 0.053 0.000 1.335 0.877 0.123 0.000 1.288 0.781 0.219 0.000 1.360 0.471 0.529 0.000 0.868 0.572 0.428 0.000 1.003 0.704 0.296 0.000 1.035 0.641 0.359 0.000 1.068 1.000 0.000 0.000 1.220 0.755 0.000 0.245 0.571 0.552 0.000 0.448 0.241 0.622 0.175 0.203 0.535 0.689 0.000 0.000 1.162 1. Using a graphical analysis, show that the proposed rate equation: k(P - Pw KEQ -TA= (1+ KAPA + KĘPE + Kw, Pw)2 Fits the above data reasonaby well. The parameter Keg is the equilibrium constant for the reaction at 120 °C, based on pressure units. 2. From your anaylsis, estimate the values for the rate constants k, the equilibrium constant Keq, and the adsorption equilibrium constants KA, KE, and Kw.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these civil engineering questions
-
A piezocone test was carried out at a site, and the following readings were recorded for the cone resistance q c and sleeve friction f s . Determine the soil present at these depths. Cone resistance,...
-
Water and diethyl ether are immiscible liquids. Charged compounds dissolve in water, and uncharged compounds dissolve in ether. a. What pH would you make the water layer in order to cause both...
-
This figure shows phase 1 vapor recovery from an underground gasoline storage tank. Before phase 1 vapor recovery was developed, gasoline vapors were vented directly into the air. Now the vapor is...
-
Ari Goldstein issued $300,000 of 11%, five-year bonds payable on January 1, 2024. The market interest rate at the date of issuance was 10%, and the bonds pay interest semiannually. Requirements 1....
-
In simple terms, what is the path-goal theory of leadership? What is the leaders function in this conceptualization?
-
Amanda McNall is the corporate controller of Scott Quarry. Scott Quarry operates 12 rock-crushing plants in Scott County, Kentucky, that process huge chunks of limestone rock extracted from...
-
Mrs. Palsgraf was waiting for a train on a platform of a railroad. When a different train came into the station, two men ran to get on that train before it left the station. While one of the men...
-
Green Landscaping Inc. is preparing its budget for the first quarter of 2014. The next step in the budgeting process is to prepare a cash receipts schedule and a cash payments schedule. To that end...
-
The 2020 season was expected to be a year of partying at Cedar Fair. Its namesake theme parkCedar Pointreached its 150 th year, and its Knotts Berry Farm was prepared to celebrate its 100 th...
-
Let f(x) be a pdf and let a be a number such that, for all > 0, f(a + ) = f(a - ). Such a pdf is said to be symmetric about the point a. (a) Give three examples of symmetric pdfs. (b) Show that if X...
-
Shop Co. has sales of $11 million and cost of sales of $5.5 million. Its selling, general, and administrative expenses are $700,000 and its research and development is $1 million. It has annual...
-
The dotplot shows the distribution of the heights (in feet) of a sample of roller coasters. The five-number summary of the data is given in the following table. Sketch a boxplot of the data. Explain...
-
Consider an economy described by the following equations: where Y is GDP, C is consumption, I is investment, G is government purchases, T is taxes, and r is the interest rate. If the economy were at...
-
Graph the expense and revenue functions. a. Determine an appropriate maximum horizontal-axis value. b. Determine an appropriate maximum vertical-axis value. c. Sketch the graphs of the expense and...
-
For a lossless line, at SIL, the voltage profile is ________, and the real power delivered, in terms of rated line voltage \(\mathrm{V}\) and surge impedance \(\mathrm{Z}_{c}\), is given by ________.
-
For equivalent \(\pi\) circuits of lossless lines, the \(A\) and \(D\) parameters are pure ________, whereas \(B\) and \(C\) parameters are pure ________.
-
48) Following the global financial crisis in 2008-2009, the economy of Greece fell apart and has struggled to regain strength. What is one reason for the demise of the Greek economy? A) Closing...
-
An educational researcher devised a wooden toy assembly project to test learning in 6-year-olds. The time in seconds to assemble the project was noted, and the toy was disassembled out of the childs...
-
As long as the pH is greater than __________, more than 50% of a protonated amine with a pK a of 10.4 will be in its neutral, nonprotonated form.
-
Explain the order of elution (with a buffer of pH 4) of each of the following pairs of amino acids on a column packed with Dowex 50 (Figure 23.3): a. Aspartate before serine b. Glycine before alanine...
-
How could you prepare the following compounds using a starting material containing no more than three carbon atoms? a. b. CH3 CH CH CHCHCH O Br CH CH2CH2CCH3
-
There is another possible explanation for purchased goodwill appearing in a sole proprietor's statement of financial position. What do you think it might be?
-
Why do the assets need to be revalued in these cases? The business has not been sold.
-
The shown partners have always shared profits and losses in the ratio: Holt 4; Stott 2: Young 1. From 1 January the assets were to be revalued as the profit sharing ratios are to be altered soon. The...

Study smarter with the SolutionInn App


