3. What is the average specific heat capacity of the unknownmetal for the three trials? 4. What...
Fantastic news! We've Found the answer you've been seeking!
Question:
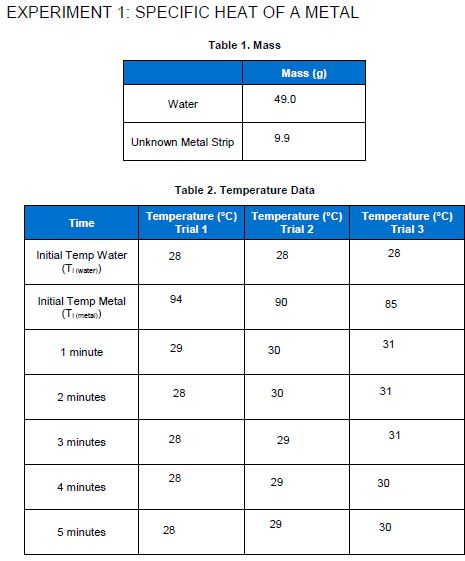
3. What is the average specific heat capacity of the unknownmetal for the three trials?
4. What is the unknown metal in Experiment 1? Use Table 1 (inthe Introduction) for reference.
5. A metal sample weighing 43.5 g at a temperature of 100.0 ?Cwas placed in 39.9 g ofwater in a calorimeter at 25.1?C. Atequilibrium, the temperature of the water and metalwas33.5?C.a.What was ?T for the water? (?T = Tfinal - Tinitial)b.What was ?T for the metal?
6. Using the specific heat of water (4.184 J/g?C), calculate howmuch heat flowedinto the water. (Show all your work for thiscalculation)
7. Calculate the specific heat of the metal. (Show all your workfor this calculation
Related Book For 

Posted Date:




