Question: 3.27 External conversion efficiency The total light output power, called the radiant flux, from a number of commercial LEDs is listed in Table 3.8.
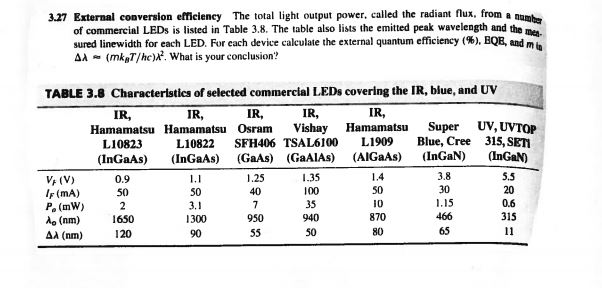
3.27 External conversion efficiency The total light output power, called the radiant flux, from a number of commercial LEDs is listed in Table 3.8. The table also lists the emitted peak wavelength and the mea sured linewidth for each LED. For each device calculate the external quantum efficiency (%), BQB, and min AA(mkaT/hc). What is your conclusion'? TABLE 3.8 Characteristics of selected commercial LEDs covering the IR, blue, and UV IR, IR, IR, IR, IR, Hamamatsu Hamamatsu Osram Vishay Hamamatsu L1909 L10823 L10822 SFH406 TSAL6100 (InGaAs) (InGaAs) (GaAs) (GaAIAs) (AIGaAs) VF (V) IF (mA) P. (mW) A (nm) A (nm) 0.9 50 2 1650 120 1.1 50 3.1 1300 90 1.25 40 7 950 55 1.35 100 35 940 50 1.4 50 10 870 80 Super UV, UVTOP Blue, Cree 315, SETI (InGaN) (InGaN) 3.8 30 1.15 466 65 5.5 20 0.6 315 11
Step by Step Solution
3.34 Rating (157 Votes )
There are 3 Steps involved in it
Answer We Put EQE m next know hext and that to hext m Ad values L10823 0053 211 531 conclusion ... View full answer

Get step-by-step solutions from verified subject matter experts


