Napthalene, C10H8, consists of two fused 6- membered rings and features a T-system that houses 10...
Fantastic news! We've Found the answer you've been seeking!
Question:
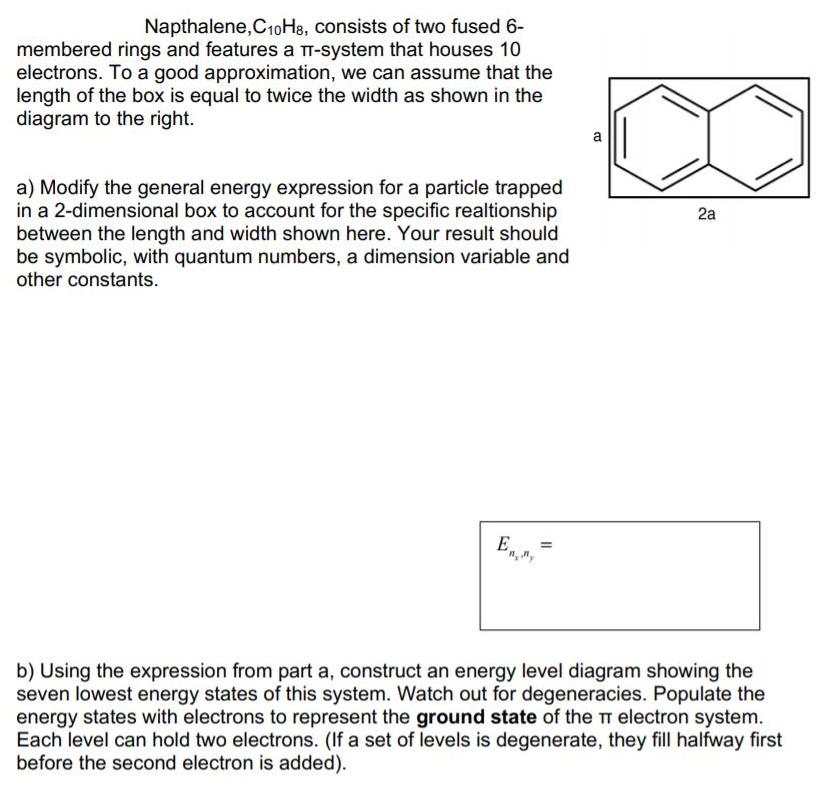

Transcribed Image Text:
Napthalene, C10H8, consists of two fused 6- membered rings and features a T-system that houses 10 electrons. To a good approximation, we can assume that the length of the box is equal to twice the width as shown in the diagram to the right. a) Modify the general energy expression for a particle trapped in a 2-dimensional box to account for the specific realtionship between the length and width shown here. Your result should be symbolic, with quantum numbers, a dimension variable and other constants. E₁, ₂ = b) Using the expression from part a, construct an energy level diagram showing the seven lowest energy states of this system. Watch out for degeneracies. Populate the energy states with electrons to represent the ground state of the TT electron system. Each level can hold two electrons. (If a set of levels is degenerate, they fill halfway first before the second electron is added). B 2a 4c) If the dimension "a" = 280 pm in the above drawing, what wavelength of light will be required to excite the lowest energy TT* transition of napthalene? (This process will move an electron from the highest occupied energy level to the lowest, unoccupied energy level.) Napthalene, C10H8, consists of two fused 6- membered rings and features a T-system that houses 10 electrons. To a good approximation, we can assume that the length of the box is equal to twice the width as shown in the diagram to the right. a) Modify the general energy expression for a particle trapped in a 2-dimensional box to account for the specific realtionship between the length and width shown here. Your result should be symbolic, with quantum numbers, a dimension variable and other constants. E₁, ₂ = b) Using the expression from part a, construct an energy level diagram showing the seven lowest energy states of this system. Watch out for degeneracies. Populate the energy states with electrons to represent the ground state of the TT electron system. Each level can hold two electrons. (If a set of levels is degenerate, they fill halfway first before the second electron is added). B 2a 4c) If the dimension "a" = 280 pm in the above drawing, what wavelength of light will be required to excite the lowest energy TT* transition of napthalene? (This process will move an electron from the highest occupied energy level to the lowest, unoccupied energy level.)
Expert Answer:
Answer rating: 100% (QA)
Solution The total energy for a Enx my Where general ... View the full answer

Posted Date:
Students also viewed these chemical engineering questions
-
As an approximation we can assume that proteins exist in the native state and in the denatured state. The standard molar enthalpy and the entropy of the denaturation of a certain protein are 512 kj /...
-
Can a transformer be used, as shown in the diagram below, to step up the voltage of a battery? Explain. Q30 Diagram
-
An electron in a one- dimensional box requires a wavelength of 8080 nm to excite an electron from the n = 2 to the n = 3 energy level. Calculate the length of this box.
-
To Premium From Actuarial Rating Advertising Sales Actuarial Premium 1e% 25% 15 60 The direct operating costs of the departments (including both variable and fixed costs) are Actuarial Premium rating...
-
Obtain Marriotts 2009 10-K through the Investor Relations portion of their website. (Using a search engine, search for: Marriott investor relations.) Once at the Investor Relations part of the...
-
Indicate whether the following statements about the conceptual framework are true or false. If false, provide a brief explanation supporting your position. (a) The fundamental qualitative...
-
In problem 3-13, you were asked to prepare a statement of cash flows for New Zealand Dairy Company. Use the information given in the problem to compute the firms free cash flows and the financing...
-
(Multiple Choice) 1. What are the components of the net position section of a governmental hospitals balance sheet? a. Unrestricted; temporarily restricted; permanently restricted b. Assets limited...
-
6. 7. 8. If one of the diameters of the circle x + y-2x-6y+6=0 is a chord of another circle 'C', whose centre is at (2, 1), then its radius is Let Bi (i = 1, 2, 3) be three independent events in a...
-
After Susan Wong graduated from State University with a degree in management science, she went to work for a computer systems development firm in the Washington, DC, area. As a student at State,...
-
The following adjustment information assuming an April 30, 2019 year end is: 1. Salaries of $5,200 have been earned by employees for the last half of April 2019. Payment by the company will be made...
-
In assessing the value of a case, an attorney a. may consult trial reporters and professional journals. b. generally, ignores the reputation of opposing counsel and the adjustor. c. must set aside...
-
Under the ____________ ____________ theory, beneficiaries of charitable organizations waive their right to sue when they accept the benefits of a charitable organization.
-
True Or False Inflation has no effect on the discounting of present-value awards.
-
If a plaintiff can raise the issue of ____________ damages, they can show the wealth of the defendant.
-
Why is proximate cause sometimes referred to as legal cause?
-
Due to a consistent decline in profits, Joe has been tasked by his supervisor to develop a plan for downsizing a specific area of the company. To be most effective, Joe's plan should include all of...
-
What is the shape of the exponential distribution?
-
Which of the following function(s) is/are carried out by piRITS or piRISC? a. Inhibits transcription of TEs b. Causes the degradation of TE RNA c. Causes chromosome breakage d. Both a and b are...
-
Scientists propose that the first macromolecules in protobionts were a. DNA molecules. b. RNA molecules. c. proteins. d. all of the above.
-
In the CRISPR-Cas system, what does tracrRNA bind to? a. crRNA and Cas1 protein b. crRNA and Cas2 protein c. crRNA and Cas9 protein d. Cas1 and Cas2 proteins

Study smarter with the SolutionInn App

