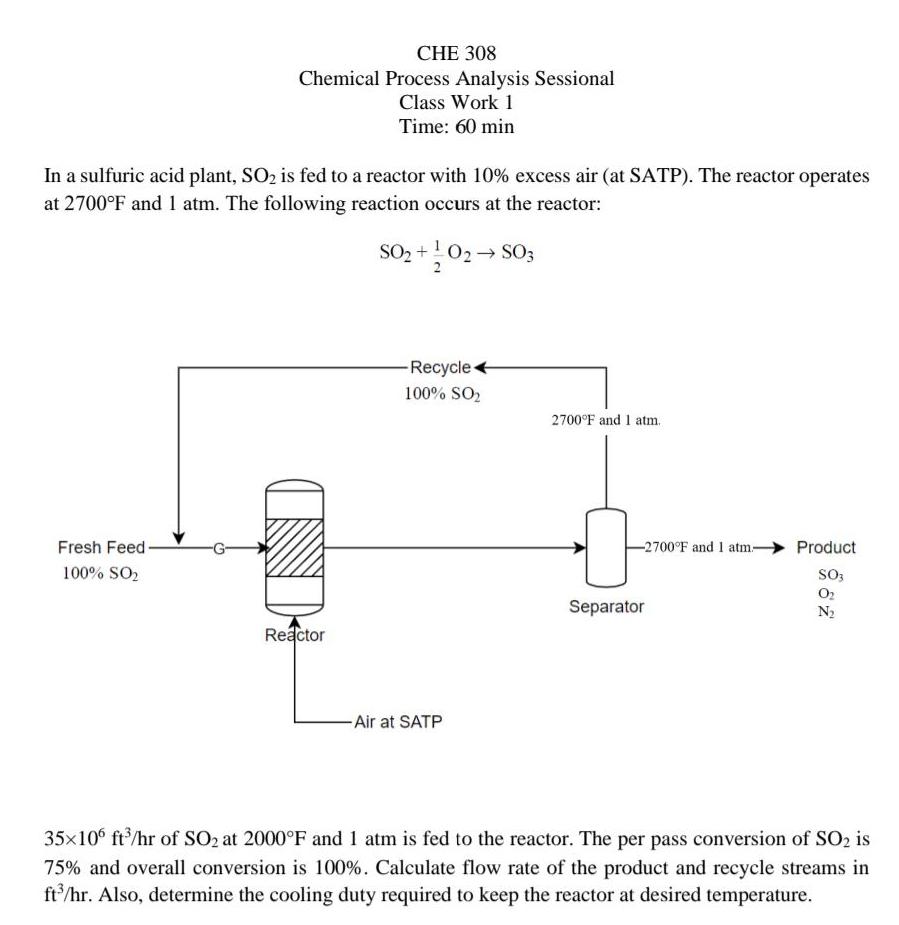
CHE 308 Chemical Process Analysis Sessional Class Work 1 Time: 60 min In a sulfuric acid...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
CHE 308 Chemical Process Analysis Sessional Class Work 1 Time: 60 min In a sulfuric acid plant, SO2 is fed to a reactor with 10% excess air (at SATP). The reactor operates at 2700°F and 1 atm. The following reaction occurs at the reactor: SO2 +!02 SO3 Recycle + 100% SO2 2700°F and 1 atm. Fresh Feed- -2700°F and 1 atm- Product 100% SO, So3 Separator O2 N2 Reactor -Air at SATP 35x10° ft/hr of SO2 at 2000°F and 1 atm is fed to the reactor. The per pass conversion of SO2 is 75% and overall conversion is 100%. Calculate flow rate of the product and recycle streams in ft'/hr. Also, determine the cooling duty required to keep the reactor at desired temperature. CHE 308 Chemical Process Analysis Sessional Class Work 1 Time: 60 min In a sulfuric acid plant, SO2 is fed to a reactor with 10% excess air (at SATP). The reactor operates at 2700°F and 1 atm. The following reaction occurs at the reactor: SO2 +!02 SO3 Recycle + 100% SO2 2700°F and 1 atm. Fresh Feed- -2700°F and 1 atm- Product 100% SO, So3 Separator O2 N2 Reactor -Air at SATP 35x10° ft/hr of SO2 at 2000°F and 1 atm is fed to the reactor. The per pass conversion of SO2 is 75% and overall conversion is 100%. Calculate flow rate of the product and recycle streams in ft'/hr. Also, determine the cooling duty required to keep the reactor at desired temperature.
Expert Answer:
Answer rating: 100% (QA)
Ansuen t So2 s fed to de actor with 1o Pxcess ay Pracar oper... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
The equilibrium constant for the reaction SO3 SO2 + O has the following values: Determine the average heat of dissociation using graphical method. T 800 K 900 K 1000 K 1105K | 0.0319 | 0.153 | 0.540...
-
Air at 1 atm, 60 F is compressed to 4 atm, after which it is expanded through a nozzle back to the atmosphere. The compressor and the nozzle are both reversible and adiabatic and kinetic energy...
-
A chemical reactor process whose production rate is a function of catalyst addition is shown in block diagram form in Figure P10.7 [10]. The time delay is T = 50 s, and the time constant T is...
-
Find the eccentricity and the distance from the pole to the directrix of the conic. Then identify the conic and sketch its graph. Use a graphing utility to confirm your results. r = 6 2 + cos 0
-
The driver of a pickup truck going 100 km/h applies the brakes, giving the truck a uniform deceleration of while it travels 20.0 m. (a) What is the speed of the truck in kilometers per hour at the...
-
Inside a NASA test vehicle, a 3.50-kg ball is pulled along by a horizontal ideal spring fixed to a friction-free table. The force constant of the spring is 225 N/m. The vehicle bas a steady...
-
How to submit a claim to private arbitration?
-
In early 20X1, SpaceTel Communications, a U.S.-based international telephone communications company, purchased the controlling interest in Sofia Telecom, Ltd. (STL) in Bulgaria. A key productivity...
-
You have been asked to prepare a December cash budget for Ashton Company, a distributor of exercise equipment. The following information is available about the company's operations: a. The cash...
-
Determine the amount of the completed gift, if any, arising from each of the following occurrences. a. A parent sells real estate valued at $1.8 million to an adult child, who pays $1 million in...
-
The total amount the borrower would need to repay a loan Question 7Select one: a. Compound value b. Present value c. Principal d. Maturity value
-
TRAINING AT THE SPICES FACTORY Ana Maria is an employee at the Mak Long Spice Factory Sdn Bhd, in Butterworth, Penang. Ana Maria has five years of experience in Mak Long's Date Spice Division....
-
Write C++ program to find a largest value among three numbers. Correct the code. include using namespace std; int main [] { } for (int n=10; n>0; n--) { cout >> n < < ", ; Cout.print < < "FIRE!/n";...
-
What inventory system involves manually counting the number of each product on hand? Explain in details.
-
Estimate the income elasticity of revenue for the motor vehicle fuel tax and evaluate the long run growth potential of the tax. Examine the years when the motor vehicle fuel tax rate was increased...
-
(i) Determine the largest internal normal N and shear V forces in the bar shown below. Enter your answers as positive numbers, and in a numerical format without using commas or powers. F N = V = 3 kN...
-
What does the study "Reexamining english only in the ESL Classroom" by Elsa Auerbach say about langauge barriers for effective teaching ?
-
Differentiate. y = ln(3x + 1) ln(5x + 1)
-
Although nitrogen dioxide (NO2) is a stable compound, there is a tendency for two such molecules to combine to form dinitrogen tetroxide (N2O4). Why? Draw four resonance structures of N2O4, showing...
-
Write structural formulas for the following organic compounds: (a) 3-methylhexane. (b) 1,3,5-trichlorocyclohexane. (c) 2,3-dimethylpentane. (d) 2-bromo-4-phenylpentane. (e) 3,4,5-trimethyloctane.
-
Two atoms have the electron configurations 1s22s22p6 and 1s22s22p63s1. The first ionization energy of one is 2080 kJ/mol, and that of the other is 496 kJ/mol. Match each ionization energy with one of...
-
What is thermodynamics?
-
Explain the scope and limitations of thermodynamics.
-
An astronaut weighs \(800 \mathrm{~N}\) on the earth's surface where the acceleration of gravity is \(g=9.83 \mathrm{~m} / \mathrm{s}^{2}\). The mass and the radius of the moon are \(7.4 \times...

Study smarter with the SolutionInn App


