Find the maximum value of the formula below 12 2|PQP-|P,Q -|P,QF 2 ++ = 4 te...
Fantastic news! We've Found the answer you've been seeking!
Question:
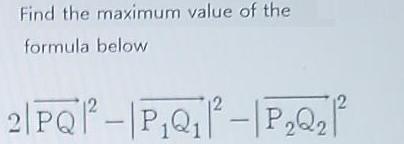
Transcribed Image Text:
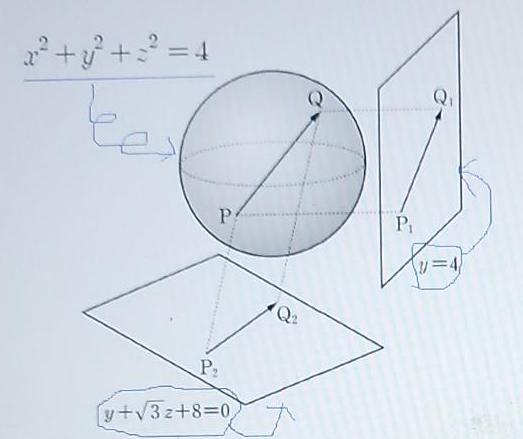
Find the maximum value of the formula below 12 2|PQP-|P,Q -|P,QF 2 ++ = 4 te Ps P1 P2 y+V3z+8%30 Find the maximum value of the formula below 12 2|PQP-|P,Q -|P,QF 2 ++ = 4 te Ps P1 P2 y+V3z+8%30
Expert Answer:
Posted Date:
Students also viewed these mathematics questions
-
Find the maximum value of the quadratic functions (a) -x2 + 3xv - 5y2 - x 4- 1. (b) - 2x2 4- 6xy - 3y2 + 4x - 3y.
-
Find the maximum value of z = xy2 subject to 2x2+y2=600; x 0, y 0.
-
Find the maximum value of z = x2y subject to x + y = 6, x 0, y 0.
-
A man drops a stone from a high bridge and hears it strike the water below exactly 4 s later. (a) Estimate the distance to the water based on the assumption that the travel time for the sound to...
-
The Wilcox Student Health Center has just implemented a new computer system and service process to improve efficiency. The process flowchart and analysis framework is also provided. As pharmacy...
-
The treasury stock in Question 14 is resold for $400,000. (a) What is the effect on the corporations revenue of the period? (b) What is the effect on stockholders equity?
-
A 1000-kg compact car and a \(2000-\mathrm{kg}\) van, each traveling at \(25 \mathrm{~m} / \mathrm{s}\), collide head-on and remain locked together after the collision, which lasts 0.20 s. (a)...
-
Record the following transactions (both budgetary and actual entries) in the General Ledger of a CPF of Santiago County. Reflect all required accruals. 1. The county issues $3,000,000 of 5%, 9-month...
-
5. Identify the type of each of the following reactions stating reason for your answers: (a) Fe2O3 + 2Al 6. Al2O3 + 2Fe + heat (b) Pb(NO3)2 + 2KI PbI2 + 2KNO heat (c) ZnCOg ZnO + CO2 In the...
-
M&S Inc. uses standard costing. The actual cost of direct labor per hour is $6.25 and the standard cost of direct labor per hour is $6.00. Two budgeted direct labor hours are allowed per finished...
-
PowerApp Corporation, a high-performance battery manufacturer, experienced strong growth in 2019 and is anticipating revenue growth in 2020 of 17%. Here is the 2019 financial picture: Memo: Growth...
-
Rank these atoms by the number of electrons they tend to lose, from fewest to most: (a) sodium, Na; (b) magnesium, Mg; (c) aluminum, Al.
-
Why does it make sense to think that the alcohol is made of very tiny particles (molecules) rather than being an infinitely continuous material?
-
Generally, what types of changes in desired saving and desired investment lead to large current account deficits in a small open economy? What factors lead to these changes in desired saving and...
-
Why does it take so much more energy to boil 10 grams of liquid water than to melt 10 grams of ice?
-
Rank these noble-gas atoms by their number of filled shells, from least to most: (a) argon, (b) radon, (c) helium, (d) neon.
-
You are working as a project manager and have identified standards and quality requirements for both the product and project. Which stage of quality management are you in? Select one: Quality...
-
An Atomic Energy Commission nuclear facility was established in Hanford, Washington, in 1943. Over the years, a significant amount of strontium 90 and cesium 137 leaked into the Columbia River. In a...
-
Because the droplets are conductors, a droplet's positive and negative charges will separate while the droplet is in the region between the deflection plates. Suppose a neutral droplet passes between...
-
You lift a book at a constant speed. Which statement is true about the work W done by your hand and the change in the book's gravitational potential energy U g ? A. W> AU>0 B. W < AU <0 C. W=AU>0 D....
-
At a point where the electric potential is 400 V, a positive charge is moving to the left. As the charge continues to move, it slows down, stops, and then begins to move back to the right. Using what...

Study smarter with the SolutionInn App


