4. Phosphoryl iodide is used in the preparation of organophosphorus derivatives and phosphate esters. Select the...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
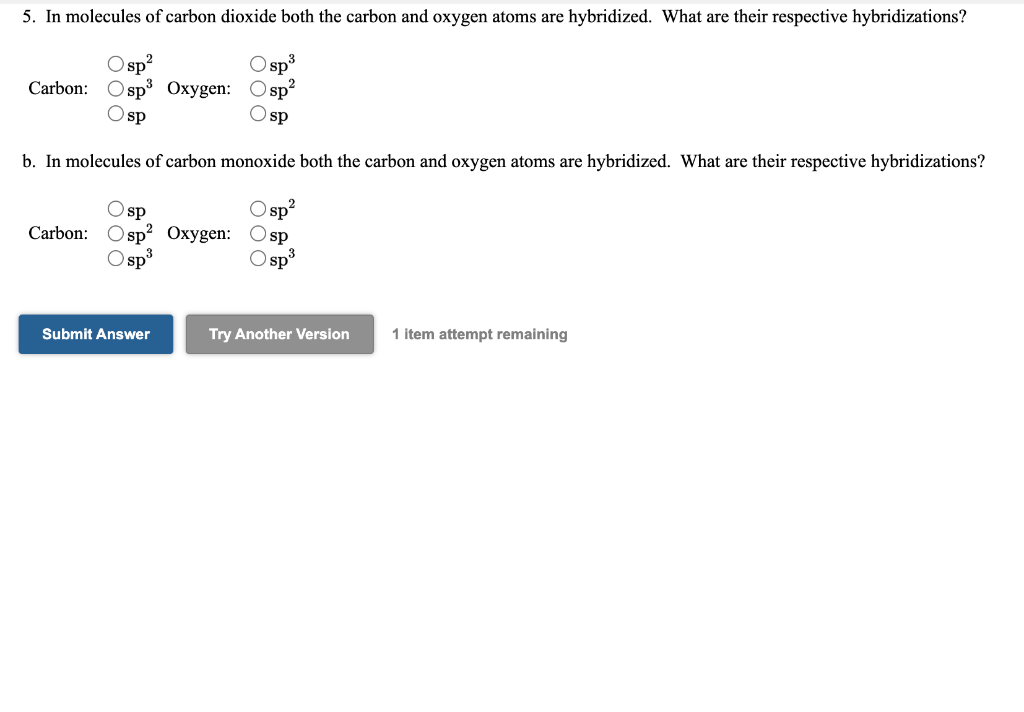
4. Phosphoryl iodide is used in the preparation of organophosphorus derivatives and phosphate esters. Select the best Lewis structure for POI3. O=P-Ï: :Ï: :Ö-P-Ï: :Ï: P=i + 5. In molecules of carbon dioxide both the carbon and oxygen atoms are hybridized. What are their respective hybridizations? Osp² Osp³ Carbon: Osp³ Oxygen: Osp² Osp Osp b. In molecules of carbon monoxide both the carbon and oxygen atoms are hybridized. What are their respective hybridizations? Osp sp² Carbon: Osp² Oxygen: Osp Osp³ Osp³ Submit Answer Try Another Version 1 item attempt remaining 4. Phosphoryl iodide is used in the preparation of organophosphorus derivatives and phosphate esters. Select the best Lewis structure for POI3. O=P-Ï: :Ï: :Ö-P-Ï: :Ï: P=i + 5. In molecules of carbon dioxide both the carbon and oxygen atoms are hybridized. What are their respective hybridizations? Osp² Osp³ Carbon: Osp³ Oxygen: Osp² Osp Osp b. In molecules of carbon monoxide both the carbon and oxygen atoms are hybridized. What are their respective hybridizations? Osp sp² Carbon: Osp² Oxygen: Osp Osp³ Osp³ Submit Answer Try Another Version 1 item attempt remaining
Expert Answer:
Answer rating: 100% (QA)
9 10 11 12 1 2 3 5 6 20 OP I 66 I 00 I OC Carbon Sp 42 15 ... View the full answer

Related Book For 

Financial Reporting and Analysis
ISBN: 978-0078025679
6th edition
Authors: Flawrence Revsine, Daniel Collins, Bruce, Mittelstaedt, Leon
Posted Date:
Students also viewed these chemistry questions
-
The following classification scheme typically is used in the preparation of a balance sheet: a. Current assets b. Investments and funds g. c. Property, plant, and equipment d. Intangible assets i....
-
The symbol W is used in the various figure parts to specify the weight of an element. If not given, assume the parts are weightless. For each figure part, sketch a free-body diagram of each element,...
-
What are the four steps involved in the preparation of a production cost report?
-
Molecular weight data for some polymer are tabulated here. Compute the following: (a) The number-average molecular weight (b) The weight-average molecular weight. (c) If it is known that this...
-
CNA Enterprises purchases an oil field and expects it to produce 1,000,000 barrels of oil. The oil field, acquired in January 2008, cost CNA $1.5 million. In 2008, the oil field produced 280,000...
-
Describe the nature of the evidence the internal revenue agent will use in the audit of Jones Company's tax return.
-
How many times can you define a function?
-
All Eyes Security Services Company provides security monitoring services. It employs four security specialists. Each specialist works an average of 180 hours a month. The companys controller has...
-
An annuity, paid in advance, is indexed to inflation at a rate of r% per period. What is the present value of this indexed annuity given a requirement of an effective investment rate of i% per period...
-
Start with the partial model in the file Ch21 P08 Build a Model.xlsx on the textbook's Web site. Kasperov Corporation has an unlevered cost of equity of 12% and is taxed at a 40% rate. The 4-year...
-
Use the brewery.xlsx file to answer the following question: How many total transactions took place at BelandAmherst? 3 5 2 7 4 2 5 8 4 4 2 5 8 5 4 2 1 9 4 3 1 5 9 3 9
-
Find all equilibrium points of the given system and determine whether we have linear stability at them or not. x' 1 - ey y' = 1- x x sin y =
-
Describe some of the benefits of e-procurement with example.
-
1 What is the primary driver when budgeting CapEx? 2 What is the primary driver when budgeting OpEx? 3 What is the primary driver when budgeting COGS? 4 Which account category is used for...
-
The stock market data of KIRI, KNR and Nifty 50 values are given below for different dates. You are required to analyse these 2 stocks from risk, return and CAPM return point of view and find the...
-
You are standing on a sheet of ice that covers the football stadium parking lot in Buffalo; there is negligible friction between your feet and the ice. A friend throws you a 0.430 kg ball that is...
-
What is a firms ROA if its operating margin is 20.7%, interest expense is 2.5% of sales, asset turnover is 1.2, and its tax rate is 19%?
-
3.16. For a system with non-identical service rates (see Sect. 3.5) and a limit of N jobs in the system (Eq. 3.13), obtain an expression for the mean service time per job, E[Ts], as a function of the...
-
Diana Gomez Corporation, a manufacturer of cowboy boots, provided the following information from its accounting records for the year ended December 31, 2014. Inventory at December 31, 2014 (based on...
-
Sea Company purchased 60% of Island Companys common stock for $180,000. On the acquisition date, Islands book value of net assets totaled $250,000 and the fair value of identifiable net assets...
-
Brower Corporation owns a manufacturing plant in the country of Oust. On December 31, 2014, the plant had a book value of $5,000,000 and an estimated fair value of $8,000,000. Ousts government has...
-
What ethical issues might arise during the fact-finding process, and how should they be handled?
-
Why are conventional files easy to design and implement?
-
Determine the business's or organization's requirements through interviews, forms, surveys, JAD, and the like, and create the appropriate models and studies for the Web site. Don't forget to consider...

Study smarter with the SolutionInn App


