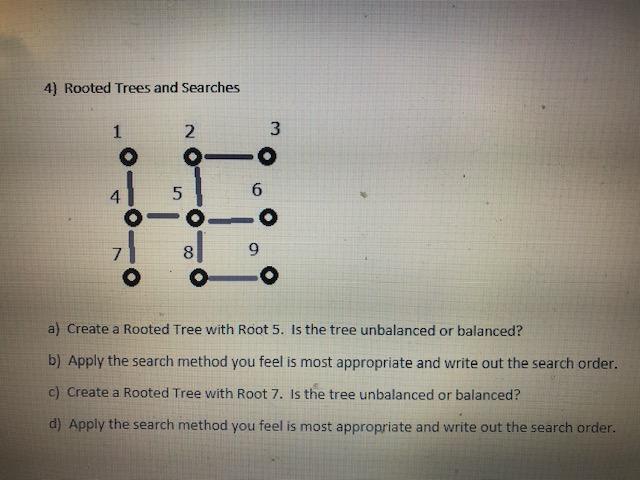
4) Rooted Trees and Searches 1 2 5 6 HE 9 3 -O a) Create a...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
4) Rooted Trees and Searches 1 2 5 6 HE 9 3 -O a) Create a Rooted Tree with Root 5. Is the tree unbalanced or balanced? b) Apply the search method you feel is most appropriate and write out the search order. c) Create a Rooted Tree with Root 7. Is the tree unbalanced or balanced? d) Apply the search method you feel is most appropriate and write out the search order. 4) Rooted Trees and Searches 1 2 5 6 HE 9 3 -O a) Create a Rooted Tree with Root 5. Is the tree unbalanced or balanced? b) Apply the search method you feel is most appropriate and write out the search order. c) Create a Rooted Tree with Root 7. Is the tree unbalanced or balanced? d) Apply the search method you feel is most appropriate and write out the search order.
Expert Answer:
Answer rating: 100% (QA)
1 A rooted tree is a tree in which the root is connected to at least one other vertex In a rooted tr... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these algorithms questions
-
YEAR BEG BAL PMT INT PRIN END BAL 1 $1,000 $360 $320 Y 2 $680 $360 $27 Z $347 3 $347 $360 $14 $347 0 TOTAL $1,081 $81 $1,000 The Value of Z in the chart (2nd year principle)?
-
On April 23, 2022, Protanica Company, a florist, purchased $112,000 of merchandise, terms were 3/15, n/45 from Botail Garden Center. Protanica returned $7,500 of damaged merchandise to Botail, which...
-
Case Study: Quick Fix Dental Practice Technology requirements Application must be built using Visual Studio 2019 or Visual Studio 2017, professional or enterprise. The community edition is not...
-
Joseph Thompson is president and sole shareholder of Jay Corporation. In December 2019, Joe asks your advice regarding a charitable contribution he plans to have the corporation make to the...
-
Let Y 1 , Y 2 , . . . , Y n be a random sample from a Pareto pdf, f Y (y; ) = /(1 + y) +1 , 0 y ; 0 < < Write f Y (y; ) in exponential form and deduce a sufficient statistic for
-
Interview an angel investor and a banker to learn what his or her expectations are when reviewing business plans for new ventures. In a two-page report, compare their criteria for choosing to fund or...
-
A manufacturer operating a standard costing system has the following data for a month: Standard Actual Number of working days 25 27 Manhours per month 5,000 5,400 Output in units 500 525 Fixed...
-
Gianna Tuck is an accountant for Post Pharmaceuticals. Her duties include tracking research and development spending in the new product development division. Over the course of the past six months,...
-
PROBLEM 3 - PARTI Clouds Corporation began operations in 2018. The year-end cost and fur values for its available for sale portfolio are as follows: Cost Fair Value December 31, 2018 $220,000...
-
Margie Frump is employed as a teacher. She occasionally purchases items that she uses in the course of her employment that are subject to depreciation. Assets held by Margie at 30 June 2021: Margie...
-
ALGO ALGO ALGO ALGO LGO GO On July 1, K. Resser opened Resser's Business Services. Resser's accountant listed the following chart of accounts: Cash Supplies Prepaid Insurance Equipment Furniture and...
-
Juanita Poblamo makes large ceramic pots for use in outdoor landscape. She currently has two models, one square and the other round. Because of the size of Juanitas creations, only one pot can be...
-
EPI educational products are currently sold without any supplemental materials. The company is considering the inclusion of instructional materials such as an overhead slide presentation, potential...
-
EPI is considering eliminating a product from its ToddleTown Tours collection. This collection is aimed at children one to three years of age and includes tours of a hypothetical town. Two products,...
-
Suppose we estimate the model y i = + u i , where u i N [ 0 , i 2 ] . (a) Show that the OLS estimator of simplifies to ^ = y . (b) Hence directly obtain the variance of y . Show that this...
-
This question presumes access to software that allows NLS and ML estimation. Consider the gamma regression model of Exercise 5-2. An appropriate gamma variate can be generated using \(y=-\lambda \ln...
-
Gotohell buys and sells regularly with customers worldwide. Its home currency is the dollar. The firm expects to receive 1.2 million in 6 months time from a customer abroad. Current exchange rates in...
-
Floyd Distributors, Inc., provides a variety of auto parts to small local garages. Floyd purchases parts from manufacturers according to the EOQ model and then ships the parts from a regional...
-
In the 2012 tax year, Michelle paid the following amounts relating to her 2010 tax return: Tax deficiency..........................................$5,000 Negligence...
-
Russell (age 50) and Linda (age 45) Long have brought you the following information regarding their income and expenses for the current year. Russell owns and operates a landscaping business called...
-
Karim Depak received a Form 1099-B showing the following stock transactions and basis during 2012: None of the stock is qualified small business stock. Calculate Karim's net capital gain or loss...
-
Unincorporated joint operation managed by one of the operators LO4 During 2018, discussions took place between Broken Bay Ltd, a company concerned with the design of specialised tools and...
-
ACCOUNTING FOR AN ASSET USED BY A NUMBER OF COMPANIES Raby Ltd and Bay Ltd are companies that have newly discovered oil wells in a MiddleEastern country. There is some distance to the nearest port...
-
Contribution of plant LO3, 4 On 1 July 2019, Tully Ltd and Cooktown Ltd signed a contractual agreement to form a joint operation for the manufacture of kitchen products. The agreement provided that...

Study smarter with the SolutionInn App


