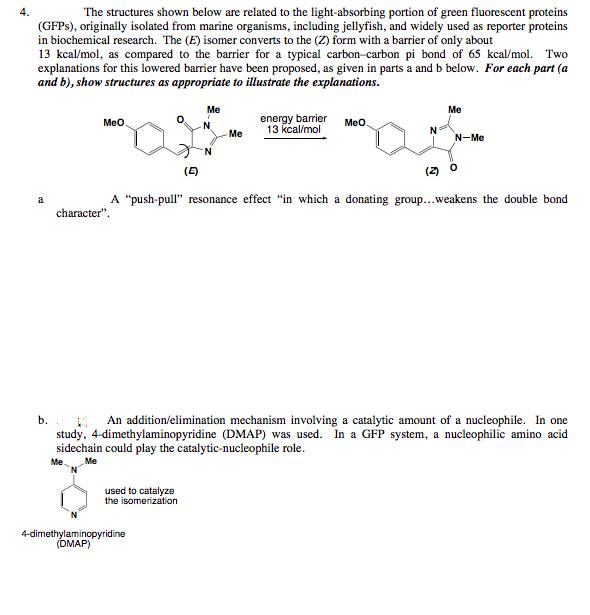
4. The structures shown below are related to the light-absorbing portion of green fluorescent proteins (GFPS),...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
4. The structures shown below are related to the light-absorbing portion of green fluorescent proteins (GFPS), originally isolated from marine organisms, including jellyfish, and widely used as reporter proteins in biochemical research. The (E) isomer converts to the (Z) form with a barrier of only about 13 kcal/mol, as compared to the barrier for a typical carbon-carbon pi bond of 65 kcal/mol. Two explanations for this lowered barrier have been proposed, as given in parts a and b below. For each part (a and b), show structures as appropriate to illustrate the explanations. Me Me energy barrier 13 kcal/mol Meo. N Meo Me N-Me (E) (2) O A "push-pull" resonance effect "in which a donating group...weakens the double bond a character". b. An addition/elimination mechanism involving a catalytic amount of a nucleophile. In one study, 4-dimethylaminopyridine (DMAP) was used. In a GFP system, a nucleophilic amino acid sidechain could play the catalytic-nucleophile role. Me Me N. used to catalyze the isomerization 4-dimethylaminopyridine (DMAP) 4. The structures shown below are related to the light-absorbing portion of green fluorescent proteins (GFPS), originally isolated from marine organisms, including jellyfish, and widely used as reporter proteins in biochemical research. The (E) isomer converts to the (Z) form with a barrier of only about 13 kcal/mol, as compared to the barrier for a typical carbon-carbon pi bond of 65 kcal/mol. Two explanations for this lowered barrier have been proposed, as given in parts a and b below. For each part (a and b), show structures as appropriate to illustrate the explanations. Me Me energy barrier 13 kcal/mol Meo. N Meo Me N-Me (E) (2) O A "push-pull" resonance effect "in which a donating group...weakens the double bond a character". b. An addition/elimination mechanism involving a catalytic amount of a nucleophile. In one study, 4-dimethylaminopyridine (DMAP) was used. In a GFP system, a nucleophilic amino acid sidechain could play the catalytic-nucleophile role. Me Me N. used to catalyze the isomerization 4-dimethylaminopyridine (DMAP)
Expert Answer:

Related Book For 

Organic Chemistry A Short Course
ISBN: 978-1111425562
13th edition
Authors: Harold Hart, Christopher M. Hadad, Leslie E. Craine, David J. Hart
Posted Date:
Students also viewed these chemistry questions
-
The costs listed below are related to a manufacturer of all-natural ice cream. In the space provided, indicate whether the cost should be classified as direct materials (DM), direct labor (DL), or...
-
2. The members of the structures shown below are made of plain concrete. The compressive strength of concrete is 4000 psi. Calculate the maximum P that can be carried by these structures. (Don't use...
-
A typical amino acid with one amino group and one carboxylic acid group, such as serine (Figure 24.18), can exist in water in several ionic forms. (a) Suggest the forms of theamino acid at low pH and...
-
The separation of operational responsibility from record keeping is meant to prevent different types of misstatements than the separation of the custody of assets from accounting. Explain the...
-
Respond to the questions in Consumer Insight 1-1. In Insight 1-1, how is the field of consumer behavior defined?
-
Four supervisors and 3 engineers are responsible for work at a construction site, and at least 2 supervisors and one engineer have to be present at all times. (a) Using two coordinates so that...
-
In Example 11.13, HENs are designed for a process involving two hot and two cold streams. Note that three designs are proposed: (1) involving only HP steam and cooling water that meets the \(N_{H X,...
-
Jenson Department Store is divided into three major departments: Mens Clothing, Womens Clothing, and Home Furnishings. Each of these three departments is supervised by a manager who reports to the...
-
The preceding section reveals that management accounting has evolved parallel with the development of technology and business environment. It is apparent that management accounting information has...
-
Chow, a PA, is assisting his client, Western Resources Inc., a closely held company, that is seeking to secure a new line of credit from a local bank. Chow has performed a review of the companys...
-
Solar Pic sells their frequency counters to various customers. 1) In contract X with the customer, Solar promises to deliver 50 frequency counters for a total price of 300,000. There is also a clause...
-
Discuss in detail the importance of DNA technology in plant breeding.
-
In Ichong v Hernandez, discuss the decision of the Supreme Court as regards whether or not RA 1180 was unconstitutional? Cite your legal basis and discuss it based on the facts as found by the Court.
-
Jayne is a Canadian who owns a hotel in Savannah, Georgia. When she brings her investment income back to Canada, how is this income classified? Explain.
-
Herbicides are usually applied in three different ways to control weeds. Name and discuss the timing of applications, and the advantages of each application in the field.
-
In general partnerships, every partner has the statutory right to participate in the management of the partnership. 1. Why are limited partnerships prohibited in the management of the limited...
-
5. Consider the state j, j2, j,m) which is a common eigenstate of the angular momentum operators J, J22, J2 and Jz where J = J +J2. Show that this state is also an eigenstate of the inner product...
-
Teasdale Inc. manufactures and sells commercial and residential security equipment. The comparative unclassified balance sheets for December 31, 2015 and 2014 are provided below. Selected missing...
-
Let us say that the (2R,3R) and (2S,3R) products in eq. 5.7 are formed in a 60:40 ratio. What products would be formed and in what ratio by adding HBr to pure (S)-3-chloro-1-butene? By adding HBr to...
-
D-Erythrose cannot exist in pyranose forms, but furanose cyclic forms are possible. Explain. Draw the structure for a-d-erythrofuranose.
-
Determine the order of reactivity for (CH3)2CHCH2Br, (CH3)3CBr, and in substitution reactions with a. Sodium cyanide. b. 50% aqueous acetone. CH CHCH2CH3 Br
-
Hannah Gilpin is the controller of Blakemore Auto Glass, a division of Eastern Glass and Window. Her division has been under pressure to improve its divisional operating income. Currently, divisions...
-
Key success factors. Dalworth Construction Company provides construction services for major projects. Managers at the company believe that construction is a people-management business, and they list...
-
How do companies add value, and what are the dimensions of performance that customers are expecting of companies?

Study smarter with the SolutionInn App


