4.1. Which two salts display discontinuities in their solubility curves? Explain what contributes to the occurrence of
Question:
4.1. Which two salts display discontinuities in their solubility curves? Explain what contributes to the occurrence of these discontinuities?
4.2. Which form of salt will be stable at a solubility of 160 kg of FeSO4 /100 kg water and 72°C?
4.3. What is the mass fraction of FeSO4 present in a saturated solution of FeSO4 at 27°C?
4.4. About 100 kg of water is added to a beaker containing 82 kg of Na2HPO4 and is continuously stirred to prepare a solution at 49°C. (use figure 1) 4.4.1. What type of solution (saturated, unsaturated, supersaturated) will be present at this temperature?
4.4.2. If the temperature of the solution is suddenly reduced to 40.5°C. How many grams of solute will precipitate out of the solution?
4.4.3. What is the chemical formula of the crystals which will form in question 4.4.2?
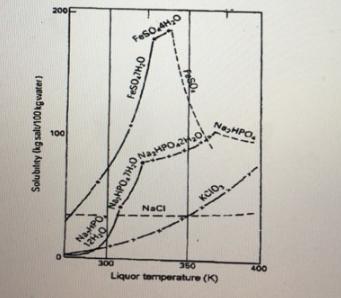
4.1. Which two salts display discontinuities in their solubility curves? Explain what contributes to the occurrence of these discontinuities? (3)
4.2. Which form of salt will be stable at a solubility of 160 kg of FeSO4 /100 kg water and 72°C? (1)
4.3. What is the mass fraction of FeSO4 present in a saturated solution of FeSO4 at 27°C? (4)
4.4. About 100 kg of water is added to a beaker containing 82 kg of Na2HPO4 and is continuously stirred to prepare a solution at 49°C. (use figure 1) 4.4.1. What type of solution (saturated, unsaturated, supersaturated) will be present at this temperature? (1)
4.4.2. If the temperature of the solution is suddenly reduced to 40.5°C. How many grams of solute will precipitate out of the solution? (3)
4.4.3. What is the chemical formula of the crystals which will form in question 4.4.2? (14.1. Which two salts display discontinuities in their solubility curves? Explain what contributes to the occurrence of these discontinuities? (3)
4.2. Which form of salt will be stable at a solubility of 160 kg of FeSO4 /100 kg water and 72°C? (1)
4.3. What is the mass fraction of FeSO4 present in a saturated solution of FeSO4 at 27°C? (4)
4.4. About 100 kg of water is added to a beaker containing 82 kg of Na2HPO4 and is continuously stirred to prepare a solution at 49°C. (use figure 1) 4.4.1. What type of solution (saturated, unsaturated, supersaturated) will be present at this temperature? (1)
4.4.2. If the temperature of the solution is suddenly reduced to 40.5°C. How many grams of solute will precipitate out of the solution? (3)
4.4.3. What is the chemical formula of the crystals which will form in question 4.4.2? (1)

Advertising & IMC Principles & Practice
ISBN: 978-0132163644
9th Edition
Authors: Sandra Moriarty, Nancy Mitchell, William Wells




