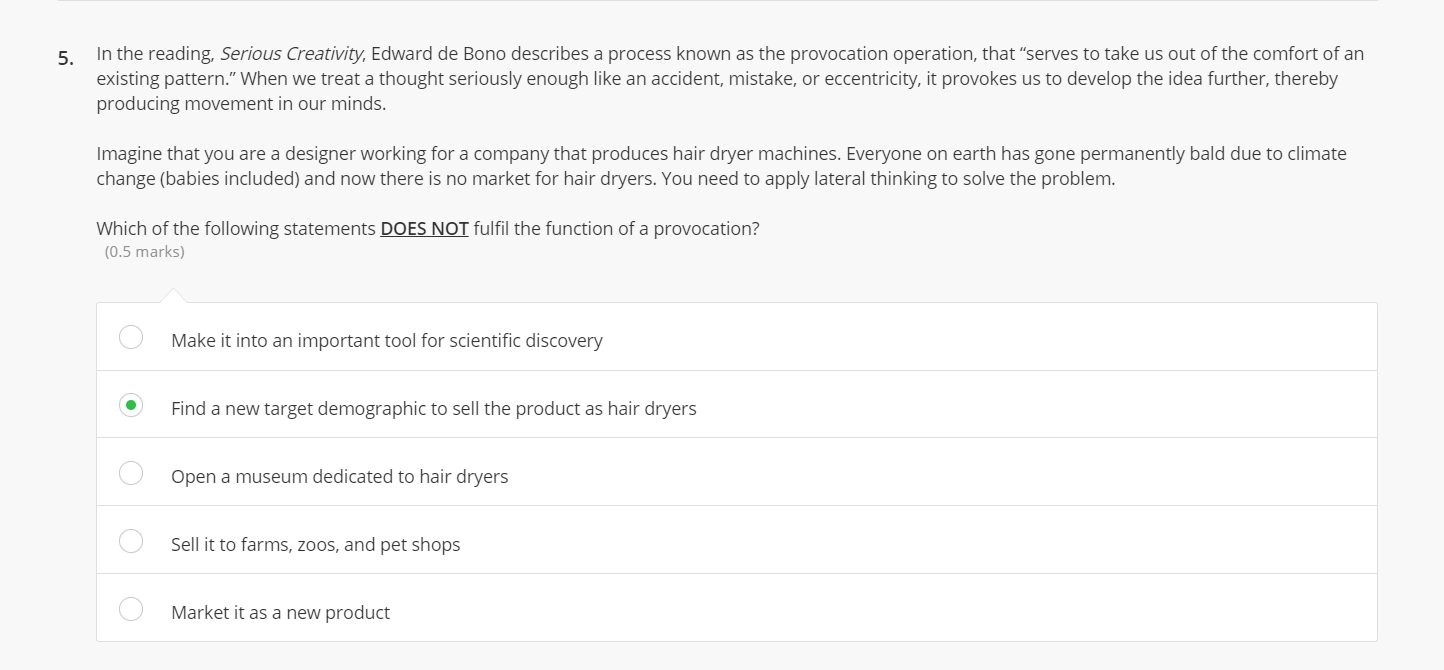
5. In the reading, Serious Creativity, Edward de Bono describes a process known as the provocation...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
5. In the reading, Serious Creativity, Edward de Bono describes a process known as the provocation operation, that "serves to take us out of the comfort of an existing pattern." When we treat a thought seriously enough like an accident, mistake, or eccentricity, it provokes us to develop the idea further, thereby producing movement in our minds. Imagine that you are a designer working for a company that produces hair dryer machines. Everyone on earth has gone permanently bald due to climate change (babies included) and now there is no market for hair dryers. You need to apply lateral thinking to solve the problem. Which of the following statements DOES NOT fulfil the function of a provocation? (0.5 marks) Make it into an important tool for scientific discovery Find a new target demographic to sell the product as hair dryers Open a museum dedicated to hair dryers Sell it to farms, zoos, and pet shops Market it as a new product 5. In the reading, Serious Creativity, Edward de Bono describes a process known as the provocation operation, that "serves to take us out of the comfort of an existing pattern." When we treat a thought seriously enough like an accident, mistake, or eccentricity, it provokes us to develop the idea further, thereby producing movement in our minds. Imagine that you are a designer working for a company that produces hair dryer machines. Everyone on earth has gone permanently bald due to climate change (babies included) and now there is no market for hair dryers. You need to apply lateral thinking to solve the problem. Which of the following statements DOES NOT fulfil the function of a provocation? (0.5 marks) Make it into an important tool for scientific discovery Find a new target demographic to sell the product as hair dryers Open a museum dedicated to hair dryers Sell it to farms, zoos, and pet shops Market it as a new product
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these electrical engineering questions
-
Which of the following statements does not properly describe accounting for OPEB (other post-employment benefits) plans? Group of answer choices a. Losses related to OPEB arises from a decrease in...
-
Which of the following statements does not properly state a basic principle for reporting an accounting change? A. retrospectively apply a change in accounting principle B. prospectively account for...
-
17. Which of the following statements does not form part of the "cell theory"? a) All living organisms are made of cells. b) The cell is the basic functional unit of all living organisms. c) All...
-
Sugar (C12H22O11) is a molecular compound that stays together inwater, while NaCl and MgSO4?7H2O are ionic compounds that dissociate into cations andanions as illustrated in the NaCl example below:...
-
Assume that the real risk-free rate of return, r*, is 3 percent and it will remain at that level far into the future. Also assume that maturity risk premiums on Treasury bonds increase from 0 percent...
-
A 2.70F capacitor is charged by a 12.0-V battery. If is disconnected from the battery and then connected to an uncharged 4.00F capacitor (Fig. 17-29). Determine the total stored energy. (a) Before...
-
In the spring of 1999, Source Associates, Inc. (Source), and Conrad A. Mamajek, Inc. (CAM), entered into a joint venture to act as a middleman for the sale of polymers manufactured by Mitsui...
-
Crystal City established a capital projects fund to account for the construction of a new bridge. During the year the fund was established, the city issued bonds, signed (and encumbered) $6 million...
-
Why would you perform a silent installation? What is the purpose of a password complexity policy? Oracle offers a free download of all editions of the Oracle Database. How does the company derive...
-
This problem continues the process of preparing the Kipley Company's Employee Payroll Register for the pay period ending January 8th, 20--. In previous chapters, gross wages were computed for each...
-
Suppose that the functions u and w are defined as follows. u(x) = -2x w (x) = -x+4 Find the following. (w) (4)= (wou) (4)=
-
A large retailer obtains merchandise under the credit terms of 3/10, net 40, but routinely takes 55 days to pay its bills. (Because the retailer is an important customer, suppliers allow the firm to...
-
Ugh Inc.s net income for the most recent year was 15,485. The tax rate was 20 percent. The firm paid 3,836 in total interest expense and deducted 2,515 in depreciation expense. What was the cash...
-
Marme, Inc. has preferred stock selling for 97 percent of par that pays an annual coupon of 14 percent. What would be Marmes component cost of preferred stock?
-
Each year you receive an end of year bonus of 1500 from your employers. You are keen to save this bonus and put it towards a deposit on a house. You have identified a savings account offering 3%...
-
financial analyst forecast Safeco core growth rate for the future to be 12% Safeco is recent dividend was 1.65 what is the value of Safeco stock when the required return is 14%
-
Observe any organization (preferable the company you are currently working in or worked earlier) and identify how the organization is implementing risk management process. Explain different risk...
-
Ann hires a nanny to watch her two children while she works at a local hospital. She pays the 19 year-old nanny $125 per week for 48 weeks during the current year. a. What is the employers portion of...
-
The normal boiling point of Br2(l) is 58.8C, and its molar enthalpy of vaporization is Hvap = 29.6 kJ/mol. (a) When Br2(l) boils at its normal boiling point, does its entropy increase or decrease?...
-
(a) Draw a picture that represents a crystalline solid at the atomic level. (b) Now draw a picture that represents an amorphous solid at the atomic level?
-
A 1 M solution of Cu(NO3)2 is placed in a beaker with a strip of Cu metal. A 1 M solution of SnSO4 is placed in a second beaker with a strip of Sn metal. A salt bridge connects the two beakers, and...
-
Role of controller, role of chief financial officer. George Jimenez is the controller at Balkin Electronics, a manufacturer of devices for the computer industry. The company may promote him to chief...
-
In the following, assume that all growth and discount rates are stated in real terms. 1. Assume the Eurozone inflation-adjusted average growth in capital stock is 3.0 percent per annum into...
-
Which conclusion presented by Ryan about the top-down approach and the bottom-up approach is most likely correct? A. Conclusion 1. B. Conclusion 2. C. Conclusion 3. Use the following information to...

Study smarter with the SolutionInn App



