IR spectroscopy provides valuable information about O molecular weight O The number of H atoms O...
Fantastic news! We've Found the answer you've been seeking!
Question:
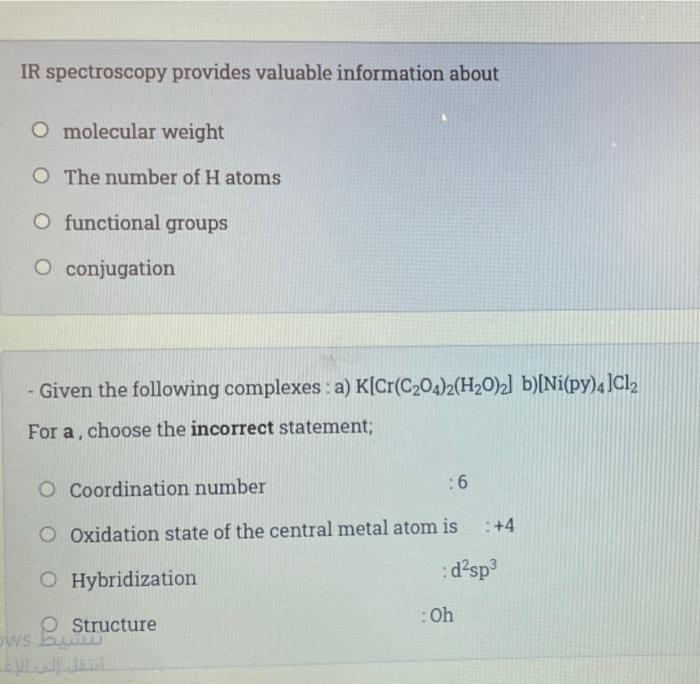
Transcribed Image Text:
IR spectroscopy provides valuable information about O molecular weight O The number of H atoms O functional groups O conjugation - Given the following complexes: a) K[Cr(C₂04)2(H₂O)2] b)[Ni(py)4]Cl₂ For a, choose the incorrect statement; O Coordination number :6 O Oxidation state of the central metal atom is : +4 O Hybridization :d²sp³ O Structure OWS b : Oh IR spectroscopy provides valuable information about O molecular weight O The number of H atoms O functional groups O conjugation - Given the following complexes: a) K[Cr(C₂04)2(H₂O)2] b)[Ni(py)4]Cl₂ For a, choose the incorrect statement; O Coordination number :6 O Oxidation state of the central metal atom is : +4 O Hybridization :d²sp³ O Structure OWS b : Oh
Expert Answer:
Answer rating: 100% (QA)
14 IR spectroscopy provides information about functional ... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemistry questions
-
Cr atoms can have a wide range of oxidation numbers. Assign oxidation numbers for the Cr atom in each compound, all of which are known compounds. a. Na2CrO4 b. Na2Cr2O7 c. CrF5 d. CrCl3 e. CrCl2
-
A hydrogen atom is in the initial state ni = n, where n > 1. (a) Find the frequency of the photon that is emitted when the electron jumps to state nf = n - 1. (b) Find the frequency of the electron's...
-
For the complexes shown here, which would have the d electron distribution shown in the diagram below: [MF6]3, [M(CN)6]3, [MF6]4, [M(CN)6]4? Note that the neutral metal atom, M, in each complex is...
-
On January 2, $217981 in 10-year, 5% bonds with a market interest rate of 9%, and interest payable semiannually, were issued for $185895. On June 30, bond interest was paid. On December 31, the...
-
THE ETHICAL DIMENSION If a Pride sales representative led the Inkels to believe that the dealership did not care about the excessive miles on the trade-in vehicle, should Pride be willing to incur...
-
Both balance sheets shown in the following table were dated as of December 31, Year 3: Required a. Based only on the information shown in the balance sheets, can Smith Co. pay a $2,000 cash dividend?...
-
How can a plaintiff enforce a settlement agreement made by the defendant?
-
Determine the value for c and the covariance and correlation for the joint probability density function fXY (x, y) = c over the range 0 < x < 5, 0 < y, and x 1 < y < x + 1.
-
Maggie s Skunk Removal Corporation s income statement listed net sales of $ 1 3 . 5 million, gross profit of $ 9 . 1 0 million, EBIT of $ 6 . 6 million, net income available to common stockholders of...
-
Gas absorption or gas scrubbing is a commonly used method for removing environmentally undesirable species from waste gases in chemical manufacturing and combustion processes. The waste gas is...
-
1. Apply OS concepts related to CPU scheduling, process synchronization, memory management and file systems to optimize the usage of system resources. 2. Use OS commands and system calls for process...
-
How many legal host addresses are possible for the network 192.168.215.224/28?
-
You are the newly hired Director of Human Resources for a city. Describe your approach to the City Manager in your first week of work ?
-
Presented below are data on three promissory notes. Determine the missing amounts. (Use 360 days for calculation.) Date of Note Terms Maturity Date Principal Annual Interest Rate 60 April 1 May 31...
-
For IESDS, it doesn't matter what order you eliminate strategies in: the same set of strategies will survive in the end (or the same unique strategy profile survives, in the case where the game is...
-
Explain about the strategy " Second Helping Strategy" and then do some work to find reliable support at least 2 for using the strategy in the Math Area. Plan your talking points on your own (why?...
-
Sunshine Airlines recently reported $7,850,00 of sales, $3,350,000 of operating costs other than depreciation, and $1,125,000 of depreciation. The company had $1,000,000 of outstanding bonds that...
-
A handrail, which weighs 120 N and is 1.8 m long. was mounted to a wall adjacent to a small set of steps (Figure P4.26). The support at A has broken, and the rail has fallen about the loose bolt at 8...
-
If Charles, a 16-year-old child model, earns $50,000 a year and is completely self supporting even though he lives with his parents, can his parents claim him as a dependent? Why or why not?...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-4. Ivan and Irene paid the following in 2012 (all by check or can otherwise be...
-
Sally hires a maid to work in her home for $250 per month. The maid is 25 years old and not related to Sally. During 2012, the maid worked 10 months for Sally. a. What is the amount of Social...
-
Cruise Canada Motorhomes Inc. (CCM) was formed on January 1, 2020, when the company issued its common shares for \(\$ 200,000\). Early in January, CCM made the following cash payments: a. For...
-
Accountants for Crowne Plaza Products Inc. have assembled the following data for the year ended December 31, 2020: {Requirement} Prepare Crowne Plaza Products's statement of cash flows using the...
-
Top managers of Culinary Imports Limited are reviewing company performance for 2020. The income statement reports a \(15 \%\) increase in net income, the fourth consecutive year showing an income...

Study smarter with the SolutionInn App


