5. Phosphate ions are abundant in cells, both as the ions themselves and as important sub-...
Fantastic news! We've Found the answer you've been seeking!
Question:
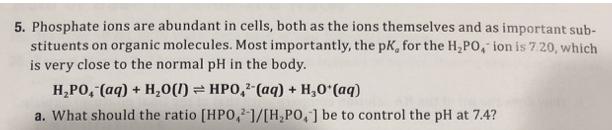
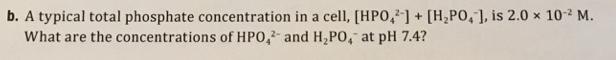
Transcribed Image Text:
5. Phosphate ions are abundant in cells, both as the ions themselves and as important sub- stituents on organic molecules. Most importantly, the pK, for the H,PO, ion is 7 20, which is very close to the normal pH in the body. H,PO, (aq) + H,0(1) = HPO, (aq) + H,0 (aq) a. What should the ratio [HPO,1/[H,PO,] be to control the pH at 7.4? b. A typical total phosphate concentration in a cell, [HPO,]+ [H,PO, ], is 2.0 x 10- M. What are the concentrations of HPO, and H,PO, at pH 7.4? 2- 5. Phosphate ions are abundant in cells, both as the ions themselves and as important sub- stituents on organic molecules. Most importantly, the pK, for the H,PO, ion is 7 20, which is very close to the normal pH in the body. H,PO, (aq) + H,0(1) = HPO, (aq) + H,0 (aq) a. What should the ratio [HPO,1/[H,PO,] be to control the pH at 7.4? b. A typical total phosphate concentration in a cell, [HPO,]+ [H,PO, ], is 2.0 x 10- M. What are the concentrations of HPO, and H,PO, at pH 7.4? 2-
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
In Prob. 346, h 0 as x 0 which cannot occur. If the maximum shear stress Ïmax due to direct shear is to be constant in this region, show that the depth h at location x is givenby 3 Ecomax 3 Fc 8...
-
Phosphate buffers are important in regulating the pH of intracellular fluids. If the concentration ratio of H2PO4- to HPO42- in a sample of intracellular fluid is 1.1; 1, what is the pH of this...
-
Phosphate buffers are important in regulating the pH of intracellular fluids at pH values generally between 7.1 and 7.2. a. What is the concentration ratio of H2PO4- to HPO4-2 in intracellular fluid...
-
The electron affinity of oxygen is -141kJ/mol, corresponding to the reaction O (g) + e- O- (g) The lattice energy of K2O(s) is 2238kJ/mol. Use these data along with data in Appendix C and Figure 7.9...
-
Blaylock Company provided the following partial comparative balance sheets and the income statement for 2016. Blaylock Company Income Statement For the Year Ended December 31, 2016 Revenues...
-
How do your profits and those of your competitor compare to the case of simultaneous decisions discussed in the text? Would you say that this example of output competition has a first mover advantage...
-
Panther Racing, LLC, is involved in competitive racing, including the development, maintenance, and operation of a racing team in the IndyCar series. Beginning in 2008, the National Guard annually...
-
Bob Night opened "The General's Favorite Fishing Hole." The fishing camp is open from April through September and attracts many famous college basketball coaches during the off-season. Guests...
-
What happened to the overhead allocation numbers? Why aren't they included?
-
Positive charges Q and Q' are placed on opposite sides of a grounded sphere of radius R at distances of 2R and 4R, respectively, from the sphere center. Show that Q' is repelled from the sphere if Q'...
-
Consider the following set of training examples: Target A B 9 8 a 8 7 a 9 10 a 7 6 a 6 5 b 11 9 b 5 7 b 4 3 9 9 a) Using 6-nearest neighbor algorithm, find the target classification for the instance...
-
Why is or is not resilience an important aspect of an organization's Information/Cyber Security program? You will need to perform an Internet or research search on resilience.
-
2. Why does Domino's carefully research supply chain potential before it decides to enter a global market?
-
Why is sustainable packaging important? How does the idea of sustainability impact the packaging value chain? How can consumers do their part to help with sustainable packaging? What are three areas...
-
Step One: Look at the 'Diversity Dimensions Wheel' and select at least three dimensions that have most impact you in life and/or work. At least one of the dimensions should be primary. Step Two:...
-
Does a strategic leader need to be a lifelong learner? Why or why not? Can a lifelong leader be a learned trait, or is it something you are born with? Explain your thoughts.
-
Given the following information, calculate the effective gross income multiplier. Sale price: $950,000 Potential Gross Income: $250,000 Vacancy and Collection Losses: 10% Miscellaneous Income: $50,000
-
Distinguish among total-moisture content, free-moisture content, equilibrium-moisture content, unbound moisture, and bound moisture.
-
Show how you would add Grignard reagents to acid chlorides or esters to synthesize the following alcohols. (a) Ph3COH (b) 3-ethyl-2-methylpentan-3-ol (c) dicyclohexylphenylmethanol
-
(a) When HBr adds across the double bond of 1,2-dimethylcyclopentene, the product is a mixture of the cis and trans isomers. Show why this addition is not stereospecific. (b) When...
-
Disiamylborane adds only once to alkynes by virtue of its two bulky secondary isoamyl groups. Disiamylborane is prepared by the reaction of BH3-THFwith an alkene. (a) Draw the structural formulas of...
-
Data from the last nine decades for the broad U.S. equity market yield the following statistics: average excess return, 8.3%; standard deviation, 20.1%. a. To the extent that these averages...
-
Suppose that the risk premium on the market portfolio is estimated at 8% with a standard deviation of 22%. What is the risk premium on a portfolio invested 25% in Toyota and 75% in Ford if they have...
-
Stock XYZ has an expected return of 12% and risk of = 1. Stock ABC has expected return of 13% and = 1.5. The markets expected return is 11%, and rf = 5%. a. According to the CAPM, which stock is a...

Study smarter with the SolutionInn App


