Question: 5. Which two D-pentose sugars give optically active alditols when reduced with NaBH4 CHO HOH CH2OH D-glyceraldehyde. CHO CHO H HOH H2OH D-erythrose HO
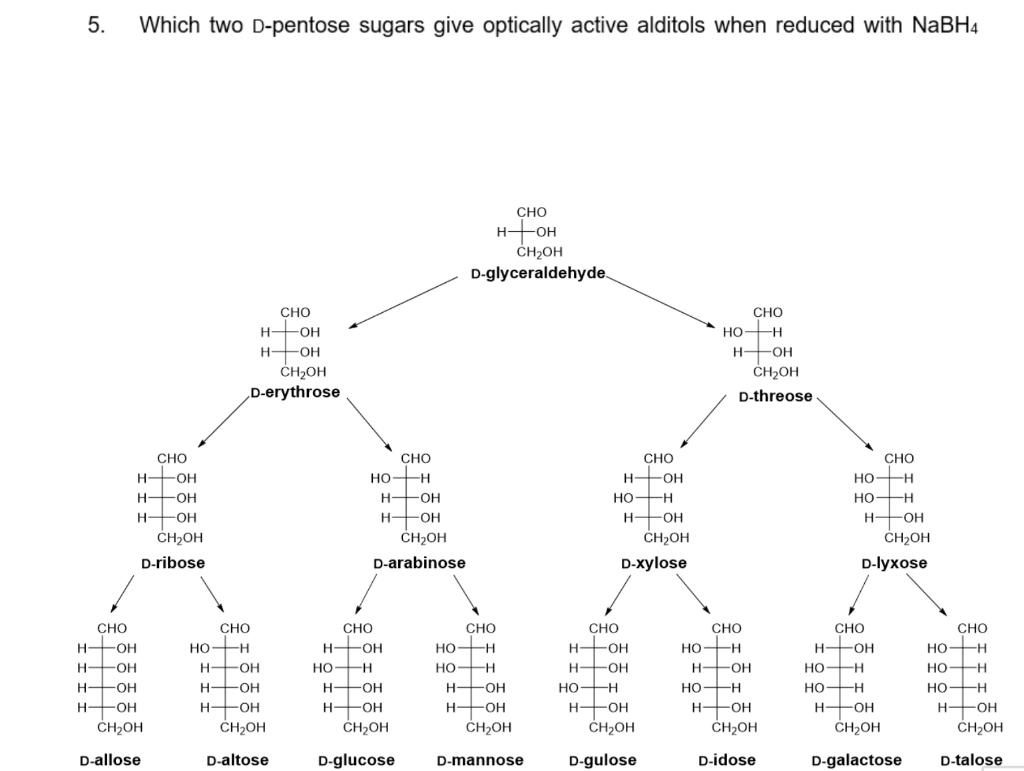
5. Which two D-pentose sugars give optically active alditols when reduced with NaBH4 CHO HOH CH2OH D-glyceraldehyde. CHO CHO H HOH H2OH D-erythrose HO H HHOH H2OH D-threose CHO CHO CHO CHO -O- HO H H -OH - -HO- H HOH -H - - HFOH CH2OH H- -HO- H FOH H -OH H2OH H2OH CH2OH D-ribose D-arabinose D-xylose D-lyxose CHO HO-H CHO CHO CHO CHO CHO CHO H FOH HO-H H FOH HO-H H FOH H OH HO-H H OH H- -O- HO H - H -OH H- OH -H HO-H HHOH H2OH HOH H +OH CH2OH H -OH -OH H FOH H -OH HHOH H2OH H- -HO- H -O- H- -OH H FOH -OH H2OH H2OH H2OH CH2OH H2OH D-allose D-altose D-glucose D-mannose D-gulose D-idose D-galactose D-talose
Step by Step Solution
3.38 Rating (160 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


