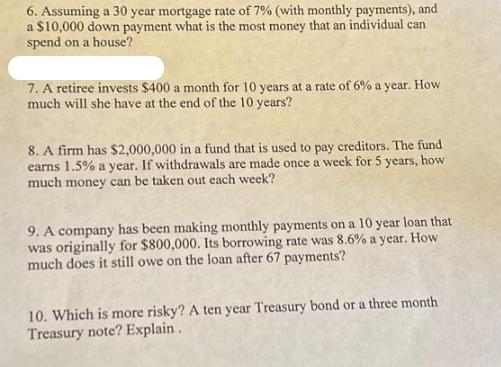
6. Assuming a 30 year mortgage rate of 7% (with monthly payments), and a $10,000 down...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
6. Assuming a 30 year mortgage rate of 7% (with monthly payments), and a $10,000 down payment what is the most money that an individual can spend on a house? 7. A retiree invests $400 a month for 10 years at a rate of 6% a year. How much will she have at the end of the 10 years? 8. A firm has $2,000,000 in a fund that is used to pay creditors. The fund earns 1.5% a year. If withdrawals are made once a week for 5 years, how much money can be taken out each week? 9. A company has been making monthly payments on a 10 year loan that was originally for $800,000. Its borrowing rate was 8.6% a year. How much does it still owe on the loan after 67 payments? 10. Which is more risky? A ten year Treasury bond or a three month Treasury note? Explain. 6. Assuming a 30 year mortgage rate of 7% (with monthly payments), and a $10,000 down payment what is the most money that an individual can spend on a house? 7. A retiree invests $400 a month for 10 years at a rate of 6% a year. How much will she have at the end of the 10 years? 8. A firm has $2,000,000 in a fund that is used to pay creditors. The fund earns 1.5% a year. If withdrawals are made once a week for 5 years, how much money can be taken out each week? 9. A company has been making monthly payments on a 10 year loan that was originally for $800,000. Its borrowing rate was 8.6% a year. How much does it still owe on the loan after 67 payments? 10. Which is more risky? A ten year Treasury bond or a three month Treasury note? Explain.
Expert Answer:
Answer rating: 100% (QA)
6 To calculate the maximum amount an individual can spend on a house we need to consider the down payment and the monthly mortgage payments Assuming a 30year mortgage rate of 7 with monthly payments w... View the full answer

Related Book For 

Posted Date:
Students also viewed these banking questions
-
Is the function {(5, 3), (6, 2), (2, 1), (7, 8), (1, 7)} one-to-one? Select Is the function {(3,8), (2, 7), (5, 6), (4, 2), (6, 7)} one-to-one? Select
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
1. Adam and Laura Jensen of Atlanta, Georgia, both of whom are in their late 20s, currently are renting an unfurnished two-bedroom apartment for $880 per month, plus $130 for utilities and $34 for...
-
A strain of E-coli Beu 397-recA441 is placed into a nutrient broth at 30° Celsius and allowed to grow. The data shown in the table are collected. The population is measured in grams and the time...
-
On July 1, 2015, Seto, Inc. purchased a fire extinguisher system for $130,000 from MTI Systems. The fire extinguisher system had an estimated life of 15 years and residual value of $2,800. Seto paid...
-
The South Dakota Department of Labor, Workers' Compensation division was sinking under a load of paper files. As a state agency which ascertains that employees are treated fairly when they are...
-
How do you delineate the differences in the System Operations Model (Figure 6.1) from its robust version (Figure 6.2)? System Development Phase Continue (5 Mission Notification Configure System for...
-
Selected comparative financial statements of Haroun Company follow. Required 1. Compute trend percents for all components of both statements using 2008 as the base year. (Round percents to one...
-
Discuss the role of process scheduling algorithms in multitasking operating systems, exploring concepts such as preemptive versus non-preemptive scheduling, priority inversion, and real-time...
-
The information listed below refers to the employees of Lemonica Company for the year ended December 31, 2016. The wages are separated into the quarters in which they were paid to the individual...
-
To prepare the upcoming party, together with your two best friends, you want to produce 100 bagel sandwiches with ham, cheese, and veggies on them. After some experimentation, you find out that the...
-
A business owned and operated by two or more persons. a. articles of incorporation b. corporation c. duty of care d. duty of loyalty e. partnership f. joint v enture g. limited liability company h....
-
Samsung must confront sunk costs. Why are sunk costs irrelevant in deciding whether to sell a product in its present condition or to make it into a new product through additional processing?
-
Consider the system of Problem 8.10-10. (a) Plot the root locus for this system, and find the value of \(K>0\) for which the system is stable. (b) Find the time constant for the system with \(K=1\)....
-
Corporations have been granted freedom of speech, freedom from unreasonable searches and seizure, and due process of law. Does it make sense that a legal (as opposed to natural) person has these...
-
How can a sole proprietor go about attracting employees who are trustworthy?
-
A middle school took all of its 6th grade students on a field trip to see a play at a theater that has 3260 seats. The students filled 95% of the seats in the theater. How many 6th graders went on...
-
Write the given system without the use of matrices. D) - ()- d (x sin t + 8 (2+ 1)
-
If the selection of cases for one sample determines the cases selected for the other sample, then the samples are ____ samples.
-
An educational psychologist was teaching a graduate seminar with seven students. He was curious to know if the amount of sleep had an impact on performance. On the first exam, he asked how many hours...
-
If a raw score is right at the mean, it has a z score of ____.
-
Calculate the entropy change when \(96 \mathrm{~g}\) of methane is heated from \(35^{\circ} \mathrm{C}\) to \(200^{\circ} \mathrm{C}\) at constant volume. Assume \(C_{V}=1.735 \mathrm{~kJ} /...
-
\(5.105 \mathrm{~mol}\) of an ideal gas is expanded from a temperature of \(300 \mathrm{~K}\) at 3 bars to \(400 \mathrm{~K}\) at 12 bars. Assume that \(C_{P}=26.73 \mathrm{~kJ} /...
-
Calculate \(\Delta S\) when \(8 \mathrm{~mol}\) of an ideal gas are heated from a temperature of 350 \(\mathrm{K}\) to a temperature of \(700 \mathrm{~K}\) at constant pressure. Assume that...

Study smarter with the SolutionInn App


