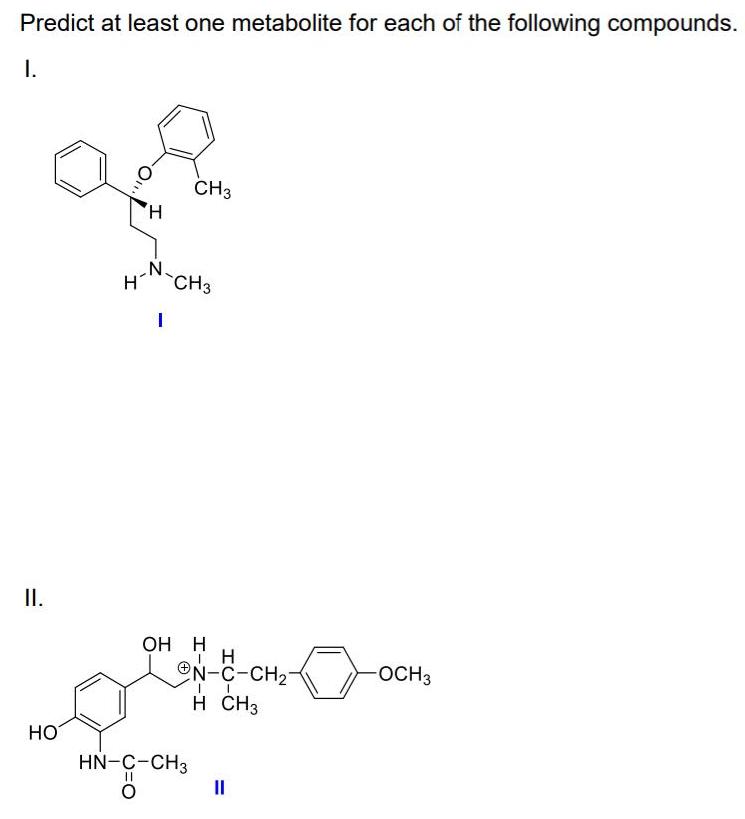
Predict at least one metabolite for each of the following compounds. I. CH3 H, N. H...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Predict at least one metabolite for each of the following compounds. I. CH3 H, N. H CH3 I. ОН Н ON-C-CH2- OCH3 H CH3 HO HN-C-CH3 %3D %3D Predict at least one metabolite for each of the following compounds. I. CH3 H, N. H CH3 I. ОН Н ON-C-CH2- OCH3 H CH3 HO HN-C-CH3 %3D %3D
Expert Answer:
Answer rating: 100% (QA)
Metabolism involves all chemical reactions that take place in the cells of living organisms as a ... View the full answer

Posted Date:
Students also viewed these chemistry questions
-
For each of the following compounds determine which (if any) lone pairs are participating in aromaticity. a. b. c. d. e. f. g. h. :N-H
-
For each of the following compounds determine whether it exhibits a cis configuration or a trans configuration or whether it is simply not stereo-isomeric. a. b. c. d. e. f. g. F
-
Predict the sites on each of the following compounds where the reaction can occur: a. CH3CH==CHOCH3 + H+ b. Cl
-
Solve Utt = cUTT 0
-
On December 31, 2014, Fenton Company sold equipment to Denver, Inc., accepting a $275,000 noninterest-bearing note receivable in full payment on December 31, 2017. Denver, Inc., normally pays 12% for...
-
What are the objectives of application controls?
-
Hostetler Energy owns 70%, Challenger Company owns 20%, and Hill Oil Company owns 10% of the working interest property 1004. Assume Hostetler Energy is the operator and incurs the following costs...
-
Digital Access Inc. needs $400,000 in funds for a project. a. With a compensating balance requirement of 20 percent, how much will the firm need to borrow? b. Given your answer to part a and a stated...
-
Suppose the manufacturer states that a bag of candies should have the following distribution of colors: 25% Red, 9% Green, 36% Yellow and 30% Brown. You open a bag that has 49 candies and count how...
-
A solution with a final volume of 500.0 mL was prepared by dissolving 25.00 mL of methanol (CH 3 OH, density = 0.791 4 g/mL) in chloroform. (a) Calculate the molarity of methanol in the solution. (b)...
-
ok k ht Product Cost NRV applied to Each Product BB $ 4,070 S 4,255 $ 4,070 FM 2,900 2,760 2,760 MB 9,486 8,772 8,772 SL 4,290 5,060 4,290 S 20,746 $ 20,847 $ 19,892 c. Prepare the appropriate...
-
1. Show how the deadlock detection algorithm works on the resource graph in Figure [1] to find all potential deadlocks. Please identify the processes and resources involved. (8 pts) R R3 P R R P
-
Blue Ridge Marketing Inc. manufactures two products, A and B. Presently, the company uses a single plantwide factory overhead rate for allocating overhead to products. However, management is...
-
To see the checking account register, do this: Multiple Choice Transactions > Chart of Accounts > double-click checking. Gear > Account and Settings. + > Single Time Activity. Create > Estimate >...
-
1) Jagadison Co. leases computer equipment to customers under sales-type leases. The equipment has no residual value at the end of the lease and the leases do not contain purchase options. Jagadison...
-
Determine if the function is continuous or not (using the definition) at the given z value. Then, Use the limit definition of the derivative to determine if the following functions are differentiable...
-
Scenario Manager Problems: Solution Have to made in Excel. Problem 1:Look into the following data: Year 1 Data Tax Rate No. of Units Sold Sales Growth Unit Price Unit Cost Discount Rate Cost Growth...
-
An auto-parts manufacturer is considering establishing an engineering computing center. This center will be equipped with three engineering workstations each of which would cost $25,000 and have a...
-
Why does an accurate calculation of EVA or profitability mean that the balance sheet will have to be restated?
-
If EPS drops after a deal, does this necessarily imply value destruction?
-
Do layoffs systematically lead to value creation?

Study smarter with the SolutionInn App

