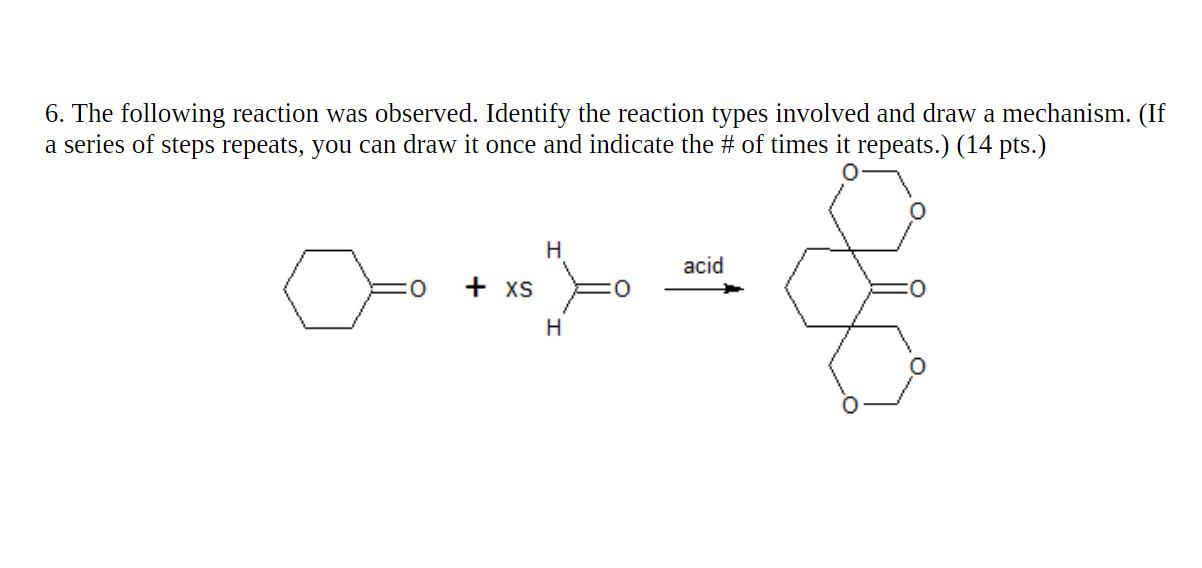
6. The following reaction was observed. Identify the reaction types involved and draw a mechanism. (If...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
6. The following reaction was observed. Identify the reaction types involved and draw a mechanism. (If a series of steps repeats, you can draw it once and indicate the # of times it repeats.) (14 pts.) acid + XS H 6. The following reaction was observed. Identify the reaction types involved and draw a mechanism. (If a series of steps repeats, you can draw it once and indicate the # of times it repeats.) (14 pts.) acid + XS H
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The following reaction was described as the cause of sulfur deposits formed at volcanic sites (see p. 780): 2H2S(g) + SO2(g) 3S(s) + 2H2O(g) It may also be used to remove SO2 from power plant stack...
-
Identify the steps involved in risk assessment. Do you think it would be effective for an organization to hire external consultants to develop its risk assessment plan?
-
The mechanism involved in the reaction catalyzed by phosphoglyceromutase is known to involve a phosphory-lated enzyme intermediate. If 3-phosphoglycerate is radioactively labeled with 32P, the...
-
The following is a comparative consolidated Statement of Financial Position for a particular firm X: Consolidated Balance Sheet (S in Millions) Current assets Cash and short-term investments Accounts...
-
Refer to Exercise 13.69. a. What is your choice of a good predictive equation? Why did you choose that particular equation? b. The model as constructed does not contain any interaction effects....
-
How are an economys production possibilities affected when workers are paid in goods rather than in cash? (World View, p. 277, about bartering in Russia.)
-
Let us assume that at time \(t\) (now), the futures price of an underlying asset is The strike of the futures option is \(K=\$ 140\), and \(F_{\text {last }}\) is the last settlement futures price...
-
The following financial data have been deter-mined from analyzing the records of Joes Ceramics (a one- product firm): Contribution margin per unit ..... $ 25 Variable cost per unit ........ $ 21...
-
There are essentially two approaches to forecasting in terms of quantitative and qualitative paradigms. Identify two techniques each (a total of 2-quant and 2-qual) techniques. Discuss "your...
-
An FRA market maker sells a EUR 100 million 3v6 FRA at a rate of 7.52%. He is exposed to the risk that interest rates will have risen by the FRA settlement date in three months' time. a. b. The...
-
The Max, is a mall-based specialty retailer of casual apparel and accessories. The company concept is to provide the customer with a focused selection of high-quality, active-oriented fashions at...
-
How are the master budgets of a retail business and a service business similar to each other? How are they different from each other?
-
Explain the double entry rule. How (if at all) does this rule change in the case of a compound entry?
-
What is a debit entry? What is a credit entry?
-
How would you describe the similarities and differences between management accounting and financial accounting? Why are they different, and why are they similar?
-
On the cash budget, why is the beginning cash balance for July the same as the beginning cash balance for the first quarter of the year? Why is the September ending cash balance the same as the first...
-
An object of mass 1.6kg experiences a force that varies with position as shown in the graph. At x = Om, the object has a velocity of 2.2m/s a) Between approximately which positions is object speeding...
-
If 2 5 9 - k 5 8 = 2 5 8 , what is the value of k?
-
Calculate the pH of the following two buffer solutions: (a) 2.0 M CH3COONa/2.0 M CH3COOH, (b) 0.20 M CH3COONa/0.20 M CH3COOH. Which is the more effective buffer? Why?
-
The concentration of commercially available concentrated sulfuric acid is 98.0 percent by mass, or 18 M. Calculate the density and the molality of the solution.
-
The enthalpy of combustion of benzoic acid (C6H5COOH) is commonly used as the standard for calibrating constant-volume bomb calorimeters; its value has been accurately determined to be -3226.7...
-
Identify from Cresseys research the six situational categories that cause nonshareable problems.
-
Which of the following is true with regard to Cresseys theory of crime causation? 1. For a fraud to occur, only one of the fraud triangle attributes (perceived pressure, perceived opportunity, or...
-
Identify which of the following is not considered a nonshareable problem from Cresseys research. 1 .Business reversal such as bankruptcy 2.Losses due to gambling 3. Justification of the fraud act by...

Study smarter with the SolutionInn App



