In the circuit shown in the figure, the capacitors are initially uncharged. The switch is first...
Fantastic news! We've Found the answer you've been seeking!
Question:
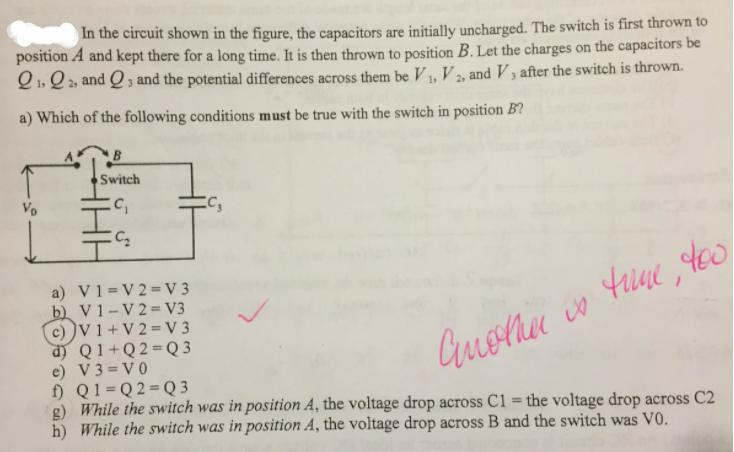
Transcribed Image Text:
In the circuit shown in the figure, the capacitors are initially uncharged. The switch is first thrown to position A and kept there for a long time. It is then thrown to position B. Let the charges on the capacitors be Q1. Q 2, and Q, and the potential differences across them be V, V2, and V, after the switch is thrown. a) Which of the following conditions must be true with the switch in position B? B Switch Vo a) V1=V 2=V 3 b) V1-V 2= V3 c) V 1+V 2 =V 3 d) Q1+Q2 Q 3 e) V3 = V0 f) Q1=Q2=Q 3 g) While the switch was in position A, the voltage drop across C1 = the voltage drop across C2 h) While the switch was in position A, the voltage drop across B and the switch was V0. %3D In the circuit shown in the figure, the capacitors are initially uncharged. The switch is first thrown to position A and kept there for a long time. It is then thrown to position B. Let the charges on the capacitors be Q1. Q 2, and Q, and the potential differences across them be V, V2, and V, after the switch is thrown. a) Which of the following conditions must be true with the switch in position B? B Switch Vo a) V1=V 2=V 3 b) V1-V 2= V3 c) V 1+V 2 =V 3 d) Q1+Q2 Q 3 e) V3 = V0 f) Q1=Q2=Q 3 g) While the switch was in position A, the voltage drop across C1 = the voltage drop across C2 h) While the switch was in position A, the voltage drop across B and the switch was V0. %3D
Expert Answer:
Answer rating: 100% (QA)
When switch is kept at A for long time then C1 and C2 will fully charged and C3 wil... View the full answer

Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Posted Date:
Students also viewed these physics questions
-
Which of the following conditions must be met before an accountant can conduct an examination of an entitys internal control? a. Management must present its assertion about the effectiveness of its...
-
In the circuit shown in Figure P33.62, assume that all parameters except for C are given. (a) Find the current as a function of time. (b) Find the power delivered to the circuit. (c) Find the current...
-
In the circuit shown in Figure P32.17, let L = 7.00 H, R = 9.00Ω, and Є = 120 V. What is the self-induced emf 0.200 s after the switch is closed? ee
-
The magnet has mass 3 . 8 5 kg and the force pulling it to the right is 1 3 6 . 8 N . When the magnet hits the floor, it continues being pulled to the right by the same magnetic force as before. The...
-
Of the education sections included in the resumes, which of the following is the most effective? a. Morehouse College, Atlanta, GA, 2001-2004. Received BA degree with a major in Business...
-
How do macroeconomic factors, such as interest rates, inflationary pressures, and systemic risk, influence the incidence and resolution of corporate insolvencies, and what are the policy implications...
-
Consider a strategy of the form \((\gamma, 0,0)\) for the investment wheel. Show that the overall factor multiplying your money after \(n\) steps is likely to be \((1+2 \gamma)^{n / 2}(1-\gamma)^{n /...
-
One item is omitted in each of the following summaries of balance sheet and income statement data for four different corporations, AL, CO, KS, and MT. Determine the missing amounts, identifying them...
-
What would be the gross debt service (GDS) ratio based on the following information? Current monthly gross income = $4000 Current monthly after-tax income = $2800 Monthly mortgage payment = $950...
-
The information necessary for preparing the 2024 year-end adjusting entries for Bearcat Personal Training Academy appears below. Bearcats fiscal year-end is December 31. 1. At the end of the year,...
-
The heat evolved in the conversion of 1 g- atom of B-sulphur into a-sulphur is 82 cal at 25C. If the specific heats ofa- and p- sulphur are 0.163 and 0.171 cal/g, then the heat evoled in the...
-
How many hours would it take for 500 mL of intravenous (IV) solution to be delivered if it is flowing at 60 drops/ minute? This set is calibrated at 100 drops per mL.
-
Evaluate the limit, if it exists., generate answer show all work. 17. (-5+ h) - 25 lim h0 h
-
Ratio evaluation is essential for a business because it enables it to analyze its financial position, liquidity, profitability, risk, solvency, efficiency, operational effectiveness, and proper use...
-
Allen has a craft store called Pentcrafto, where she sells home decor products made from recycled paper. In the context of marketing, what utilities are Pentcrafto's products most likely to provide?...
-
Determine the number of hours of direct labor required per $3000 worth of raw materials: Labor hours per $ 3000 = 4.5 hours Labor hours per $3000=4.5hours Find the number of $3000 increments in the...
-
Assume that the average firm in your company's industry is expected to grow at a constant rate of 6% and that its dividend yield is 8%. Your company is about as risky as the average firm in the...
-
Sue Deliveau opened a software consulting firm that immediately paid $2,000 for a computer. Was this event a transaction for the business?
-
A lead bullet moving at 200 m/s is stopped in a block of wood. Assuming that all of the energy change goes into heating the bullet, find the final temperature of the bullet if its initial temperature...
-
A block of mass 0.5 kg rests on the inclined surface of a wedge of mass 2 kg, as in figure. The wedge is acted on by a horizontal force and slides on a frictionless surface. (a) If the coefficient of...
-
FM radio stations have carrier frequencies that are separated by 0.20 MHz. When the radio is tuned to a station, such as 100.1 MHz, the resonance width of the receiver circuit should be much smaller...
-
A blender does 5000 J of work on the food in its bowl. During the time the blender runs, 2000 J of heat is transferred from the warm food to the cooler environment. What is the change in the thermal...
-
Which system contains more atoms: 5 mol of helium (A = 4) or 1 mol of neon (A = 20)? A. Helium B. Neon C. They have the same number of atoms.
-
A sample of ideal gas is in a sealed container. The temperature of the gas and the volume of the container are both increased. What other properties of the gas necessarily change? (More than one...

Study smarter with the SolutionInn App


