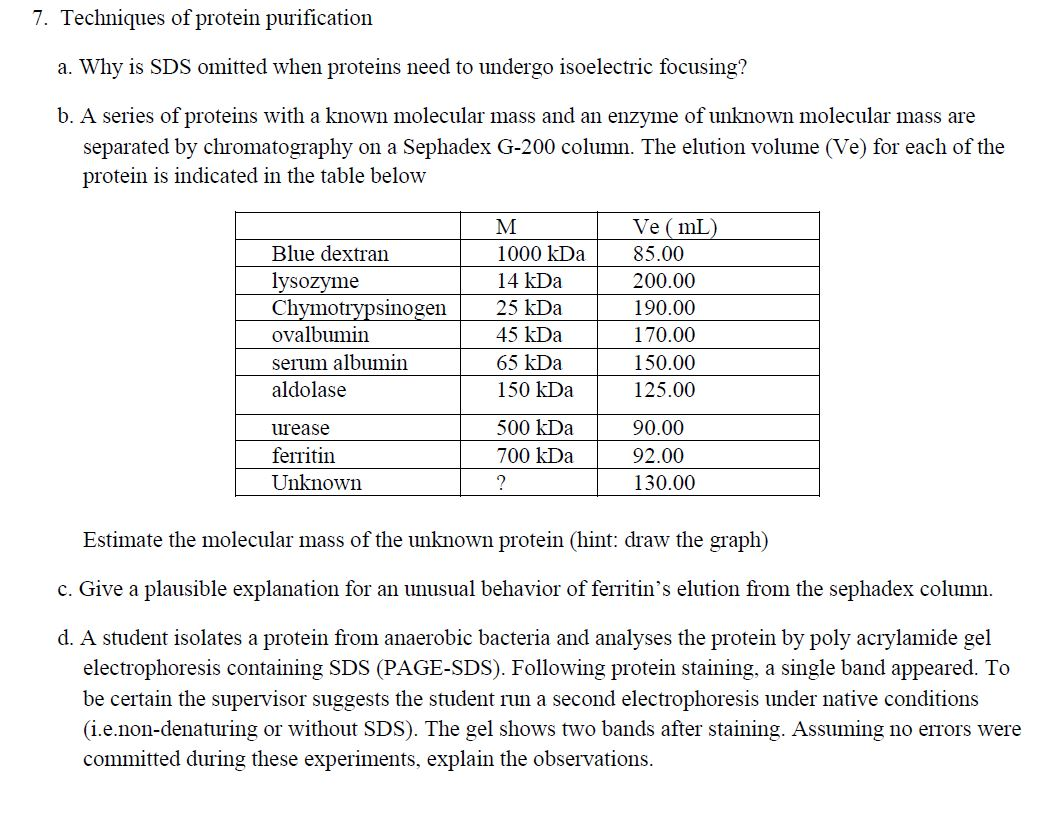
7. Techniques of protein purification a. Why is SDS omitted when proteins need to undergo isoelectric...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
7. Techniques of protein purification a. Why is SDS omitted when proteins need to undergo isoelectric focusing? b. A series of proteins with a known molecular mass and an enzyme of unknown molecular mass are separated by chromatography on a Sephadex G-200 column. The elution volume (Ve) for each of the protein is indicated in the table below Blue dextran lysozyme Chymotrypsinogen ovalbumin serum albumin aldolase urease ferritin Unknown M 1000 kDa 14 kDa 25 kDa 45 kDa 65 kDa 150 kDa 500 kDa 700 kDa ? Ve (mL) 85.00 200.00 190.00 170.00 150.00 125.00 90.00 92.00 130.00 Estimate the molecular mass of the unknown protein (hint: draw the graph) c. Give a plausible explanation for an unusual behavior of ferritin's elution from the sephadex column. d. A student isolates a protein from anaerobic bacteria and analyses the protein by poly acrylamide gel electrophoresis containing SDS (PAGE-SDS). Following protein staining, a single band appeared. To be certain the supervisor suggests the student run a second electrophoresis under native conditions (i.e.non-denaturing or without SDS). The gel shows two bands after staining. Assuming no errors were committed during these experiments, explain the observations. 7. Techniques of protein purification a. Why is SDS omitted when proteins need to undergo isoelectric focusing? b. A series of proteins with a known molecular mass and an enzyme of unknown molecular mass are separated by chromatography on a Sephadex G-200 column. The elution volume (Ve) for each of the protein is indicated in the table below Blue dextran lysozyme Chymotrypsinogen ovalbumin serum albumin aldolase urease ferritin Unknown M 1000 kDa 14 kDa 25 kDa 45 kDa 65 kDa 150 kDa 500 kDa 700 kDa ? Ve (mL) 85.00 200.00 190.00 170.00 150.00 125.00 90.00 92.00 130.00 Estimate the molecular mass of the unknown protein (hint: draw the graph) c. Give a plausible explanation for an unusual behavior of ferritin's elution from the sephadex column. d. A student isolates a protein from anaerobic bacteria and analyses the protein by poly acrylamide gel electrophoresis containing SDS (PAGE-SDS). Following protein staining, a single band appeared. To be certain the supervisor suggests the student run a second electrophoresis under native conditions (i.e.non-denaturing or without SDS). The gel shows two bands after staining. Assuming no errors were committed during these experiments, explain the observations.
Expert Answer:
Answer rating: 100% (QA)
Answer 7a Isoelectric point is that pH when the proteins net charge is zero ie in electrically neutral state The purpose of the isoelectric focusing i... View the full answer

Related Book For 

Biochemistry Concepts and Connections
ISBN: 978-0321839923
1st edition
Authors: Dean R. Appling, Spencer J. Anthony-Cahill, Christopher K. Mathews
Posted Date:
Students also viewed these biology questions
-
A protein gives a single band on SDS gel electrophoresis, as shown in lanes 1 and 2 below. There is little if any effect from adding β -mercaptoethanol (BME) to the sample; if anything,...
-
When proteins are separated using native gel electrophoresis, size, shape, and charge control their rate of migration on the gel. Why does DNA separate based on size, and why do we not worry much...
-
Under ideal conditions a certain bacteria population is known to double every three hours. Suppose that there are initially 100 bacteria. (a) What is the size of the population after 15 hours? (b)...
-
Find a and b if, y= ax+b has mean 6 and variance is unity, where x is a random variable with mean 8 and variance 16.
-
Post, CPA, accepted an engagement to audit the financial statements of General Company, a new client. General is a publicly held retailing entity that recently replaced its operating management. In...
-
August 2014 bank statement and cash T-account for Martha Company follow: No deposits were in transit and no cheques were outstanding at the end of July. Required 1. Identify and list the deposits in...
-
What is the difference between a predator and a situational (accidental) fraudster?
-
In 2012, Gurney Construction Company agreed to construct an apartment building at a price of $1,200,000. The information relating to the costs and billings for this contract is shown below....
-
Compare and contrast benefits and challenges that exist between centralized database management systems and distributed database management systems. Identify potential business environments where...
-
Cardinal Company is considering a five-year project that would require a $2,975,000 investment in equipment with a useful life of five years and no salvage value . The company's discount rate is 14%....
-
Consider the binary search tree (BST) below: (10 3 20 (30 (35) 40 (36 (60) Show the sequence of nodes visited to find the predecessor of 33.
-
Question 2 10 pts Air in a hot air balloon does 37 J of work while expanding, and also absorbs 77.3 of heat from a heat source. What is the change in the balloon air's internal energy?
-
Suppose you want to increase the income component of your return from your real estate equity portfolio but you do not want to sacrifice the appreciation component. What must you look for? If you...
-
Does increasing leverage usually primarily increase the expected income component of the return or the appreciation component? Why?
-
If gross profit is 30%, then what is the markup on cost?For financial reporting of companies using LIFO, the lower of cost or market approach can be applied toFor financial reporting of companies...
-
Stone company produces electronic components. Lately, the company has been facing a drop in sales because of fierce competition and a downturn in the market. Consequently, Stone Corporation's cash...
-
Question 1 a. Bank of City has the following financial statement. What is the risk-weighted value of the assets according to BASEL? What is the ratio of risk weighted ratio (i.e., T1+ T2) (10...
-
Use multiplication or division of power series to find the first three nonzero terms in the Maclaurin series for each function. y = e x2 cos x
-
Carbon dioxide is dissolved in blood (pH 7.4) to form a mixture of carbonic acid and bicarbonate. Neglecting free CO2, what fraction will be present as carbonic acid? Would you expect a signicant...
-
Radioactive uracil can be used to label all of the pyrimidine residues in DNA. Using either names or structures, present pathways for the conversion of uracil to dTTP and to dCTP for each reaction,...
-
Te activity of an enzyme requires a glutamic acid to display its - COOH functional group in the protonated state. Suppose the pKa of the - COOH group is 4.07 a. Will the enzyme be more active at pH...
-
Write a program in \(\mathrm{R}\) that generates a sample \(X_{1}, \ldots, X_{n}\) from a specified distribution \(F\), computes the empirical distribution function of \(X_{1}, \ldots, X_{n}\),...
-
Let \(\left\{X_{n}ight\}_{n=1}^{\infty}\) be a sequence of random variables such that \(X_{n}\) has a UNI\(\operatorname{FORM}\left\{0, n^{-1}, 2 n^{-2}, \ldots, 1ight\}\) distribution for all \(n...
-
Write a program in \(\mathrm{R}\) that generates a sample from a population with distribution function \[F(x)= \begin{cases}0 & x

Study smarter with the SolutionInn App


