(7) The formation of boron trinitrate can be done by the reaction of: B:Broe) + HNOS(a4)...
Fantastic news! We've Found the answer you've been seeking!
Question:
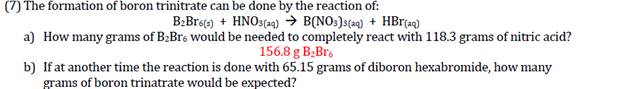
Transcribed Image Text:
(7) The formation of boron trinitrate can be done by the reaction of: B:Broe) + HNOS(a4) → B(NO:)s(ac) + HBr(aq) a) How many grams of B2Bre would be needed to completely react with 118.3 grams of nitric acid? 156.8 g B;Br. b) If at another time the reaction is done with 65.15 grams of diboron hexabromide, how many grams of boron trinatrate would be expected? (7) The formation of boron trinitrate can be done by the reaction of: B:Broe) + HNOS(a4) → B(NO:)s(ac) + HBr(aq) a) How many grams of B2Bre would be needed to completely react with 118.3 grams of nitric acid? 156.8 g B;Br. b) If at another time the reaction is done with 65.15 grams of diboron hexabromide, how many grams of boron trinatrate would be expected?
Expert Answer:
Answer rating: 100% (QA)
Oate Poge Na Given reaction H Briag first we balance the chemical equation B BYt 6HNO3 ag 2 BNOgt H8... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Can you mention some tasks that can be done by one or a combination of TCP segments?
-
Whatever can be done by a tariff can be done by a quota. Discuss.
-
What can be done by a general partner to limit the extent of its obligations to the limited partnership?
-
A group of investors is considering buying the Wheelwright Corporation, but does not want to contribute to the companys financial support after the purchase. Wheelwrights management has offered the...
-
A report released by the Pew Internet & American Life Project entitled The Internet & Consumer Choice focused on current online issues. Respondents were asked to indicate their level of agreement (1...
-
The authors of the article "A Probabilistic Insulation Life Model for Combined Thermal Electrical Stresses" (IEEE Trans. on Elect, Insulation, 1985: 519-522) state that "the Wei bull distribution is...
-
For each of the following sets of data, (1) calculate the mean of the scores \(\left(\mathrm{X}^{-} ight),(2)\) calculate the deviation of each score from the mean \(\mathrm{X}-\mathrm{X}^{-}\), and...
-
Texas Oil Company (TOC) paid $ 3,000,000 for an oil reserve estimated to hold 50,000 barrels of oil. Oil production is expected to be 10,000 barrels in year 1, 30,000 barrels in year 2, and 10,000...
-
On January 1, 2021, Entity A Company acquired all the assets and assumed all the liabilities of Entity B Company and merged Entity B into Entity A. In exchange for the net assets of Entity B, Entity...
-
The service life of a battery used in a cardiac pacemaker is assumed to be normally distributed. A random sample of ten batteries is subjected to an accelerated life test by running them continuously...
-
What is demand forecasting? What are the importance and benefits of forecasting customer demand? Provide an overview of how a bank and a furniture manufacturer would forecast demand? In your response...
-
6. In 2022, Johnson Company contracted to deliver 100 boats to another company for $1,000,000 in 2023 and 2024. Outline the 5 step revenue recognition process based on FASB ASC 606 for this...
-
CAF company is thinking about replacing a 5-year-old car with a new one. The old car has an original cost of $50,000, with a remaining book value of $10,000, a remaining life of 1 year, and a...
-
Delph Company uses a job-order costing system and has two manufacturing departments-Molding and Fabrication. The company provided the following estimates at the beginning of the year: Machine-hours...
-
The accounting records of NuTronics, Incorporated, include the following information for the year ended December 31: Inventory of materials Inventory of work in process Inventory of finished goods...
-
The following financial statement information is from five separate companies. Company A Company B Company C Company D Company E Beginning of year Assets Liabilities $ 45,000 36,900 $ 35,100 $ 28,800...
-
Analyze the T-accounts to find the direct materials used and total materials used. Raw Materials Inventory 40 215 65 Bal. Purchased Bal. Used Work-in-Process Inventory Bal. Direct Materials Direct...
-
To balance the chemical equation SiH3 + O2 SiO2 + HO, you could introduce coefficients a, b, c, d and write aSiH3 + bO2 cSiO + dHO then write linear equations for each element. The equation for Si...
-
Which of the six rules we described for ACK generation can be applied to the case where a client or a server receives a FIN segment from the other end?
-
Draw the constellation diagram for the following cases. Find the peak amplitude value for each case and define the type of modulation (ASK, FSK, PSK, or QAM).The numbers in parentheses define the...
-
In Example 11.4 (Figure 11.13) how many frames are in transit at the same time? Figure 11.13 Sending node Receiving node Network Network Data-link Data-link Legend Packet Frame 0 Start the timer....
-
In the ground state of a Fermi system, the chemical potential is identical to the Fermi energy: \((\mu)_{T=0}=\varepsilon\left(p_{F} ight)\). Making use of the energy spectrum \(\varepsilon(p)\) of...
-
Solve the Gross-Pitaevskii equation (11.2.23) in a harmonic trap for the case when the scattering length \(a\) is zero. Show that this reproduces the properties of the ground state of the...
-
Solve the Gross-Pitaevskii equation and evaluate the mean field energy, see equations (11.2.21) and (11.2.23), for an isotropic harmonic oscillator trap with frequency \(\omega_{0}\) for the case \(N...

Study smarter with the SolutionInn App


