7:38 PM Mon Dec 11 Name: 1 of 1 Date: Unit III: Nuclear Half-Life Assignment N...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
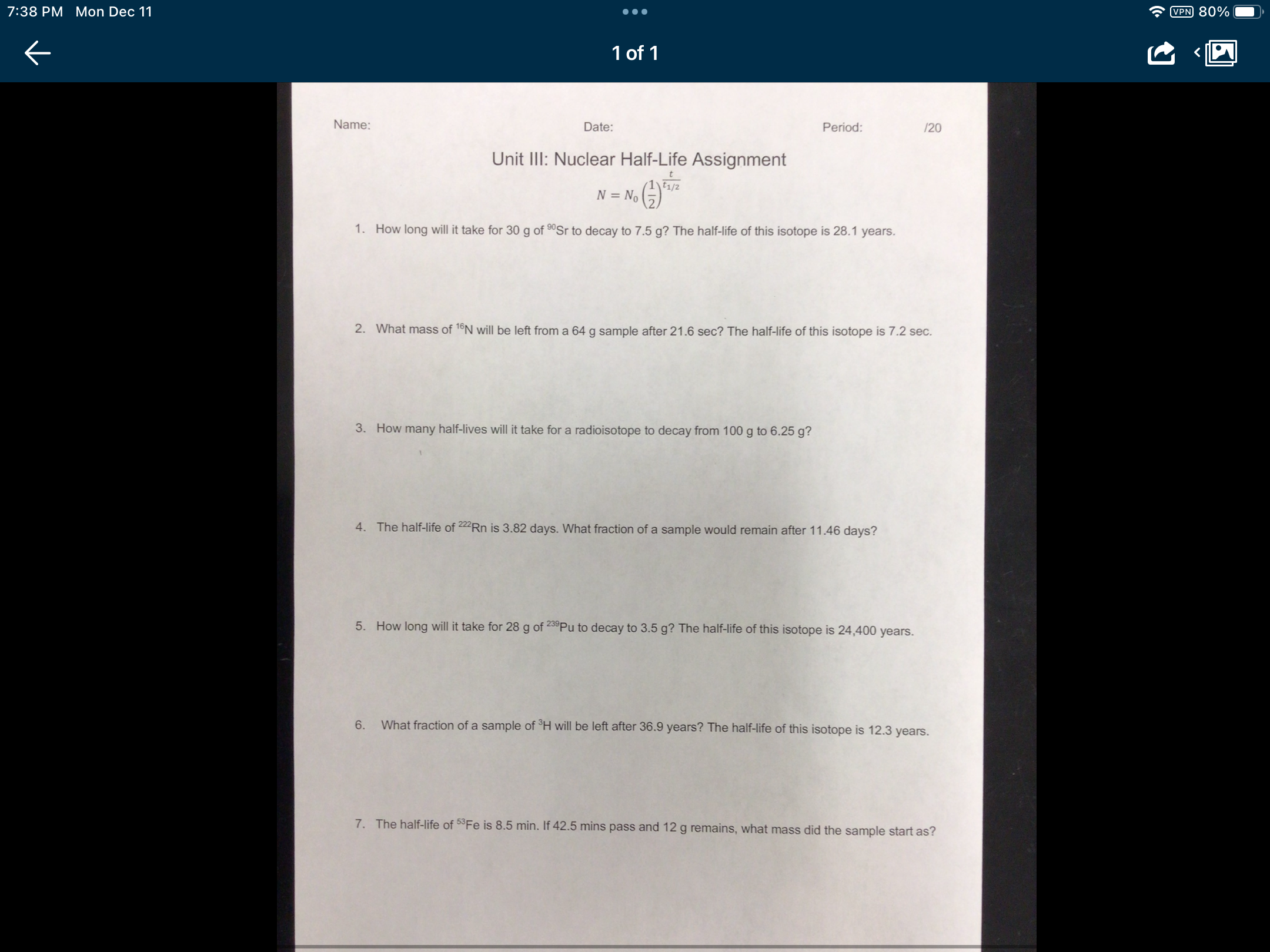
7:38 PM Mon Dec 11 Name: 1 of 1 Date: Unit III: Nuclear Half-Life Assignment N = No No 1/ Period: /20 1. How long will it take for 30 g of 90Sr to decay to 7.5 g? The half-life of this isotope is 28.1 years. 2. What mass of 16N will be left from a 64 g sample after 21.6 sec? The half-life of this isotope is 7.2 sec. 3. How many half-lives will it take for a radioisotope to decay from 100 g to 6.25 g? 4. The half-life of 222 Rn is 3.82 days. What fraction of a sample would remain after 11.46 days? 5. How long will it take for 28 g of 239Pu to decay to 3.5 g? The half-life of this isotope is 24,400 years. 6. What fraction of a sample of H will be left after 36.9 years? The half-life of this isotope is 12.3 years. 7. The half-life of 53 Fe is 8.5 min. If 42.5 mins pass and 12 g remains, what mass did the sample start as? VPN 80% 7:38 PM Mon Dec 11 Name: 1 of 1 Date: Unit III: Nuclear Half-Life Assignment N = No No 1/ Period: /20 1. How long will it take for 30 g of 90Sr to decay to 7.5 g? The half-life of this isotope is 28.1 years. 2. What mass of 16N will be left from a 64 g sample after 21.6 sec? The half-life of this isotope is 7.2 sec. 3. How many half-lives will it take for a radioisotope to decay from 100 g to 6.25 g? 4. The half-life of 222 Rn is 3.82 days. What fraction of a sample would remain after 11.46 days? 5. How long will it take for 28 g of 239Pu to decay to 3.5 g? The half-life of this isotope is 24,400 years. 6. What fraction of a sample of H will be left after 36.9 years? The half-life of this isotope is 12.3 years. 7. The half-life of 53 Fe is 8.5 min. If 42.5 mins pass and 12 g remains, what mass did the sample start as? VPN 80%
Expert Answer:

Posted Date:
Students also viewed these physics questions
-
s Alyeska Services Company, a division of a major oil company, provides various services to the operators of the North Slope oil field in Alaska. Data concerning the most recent year appear below:...
-
In 43 of the 67 years from 1950 through 2017 (in 2011 there was virtually no change), the S&P 500 finished higher after the first five days of trading. In 38 out of 43 years, the S&P 500 finished...
-
Two years ago, Capricorn Inc. purchased an Asset A for $66,000. The asset yields annual operating cash flow of $20,000 for 6 years. The company is now looking into replacing Asset A with Asset B,...
-
Is there more to marketing than building product and brand awareness than what people discuss? How do marketing managers and executives manage consistent messaging across cultures?
-
The State Conservation Department used surveillance cameras to study the reaction of white-tailed deer to traffic as they used a wildlife underpass to cross a major highway. When a car or truck...
-
This case will familiarize you with some of the assets and liabilities of that company. Use Amazon.com balance sheet to answer the following questions. Requirements 1. Which balance sheet format does...
-
The most important step in the employment process is the interview. During the interview, an employer will ask you a variety of questions about your abilities, qualifications, and attitudes. You must...
-
Draw an entity-relationship diagram for the following situation: Many students play many different sports. One person, called the head coach, assumes the role of coaching all these sports. Each of...
-
1. Assume that a 7% coupon bond with a 30 year maturity has a par value of $1,000. Assuming coupon payments are made twice a year; Determine the value of the bond to an investor whose required rate...
-
Q1-Waterbrook Farm includes 210 acres of cropland. The farm owner wishes to plant this acreage in both corn and soybeans. The profit per acre in com production is $375 and in soybeans is $365. A...
-
Is there or can there be an end to digital technology advancements?
-
Do you believe accountants and the accounting profession have been successful at adapting to digital disruption, undertaking digital business transformation, and operating in digital business...
-
Identify three or more approaches accountants and accounting leaders have taken to adapt to digital disruption.
-
How do digital technology advancements disrupt data availability? How does this disruption of data availability create competitive threats and loss of relevance risks for the accounting profession?
-
Which IFAC future-ft accounting role requires the strongest accelerated change and transformation and accelerated innovation competencies?
-
The reading speed of second grade students in a large city is approximately normal, with a mean of 88 words per minute (wpm) and a standard deviation of 10 wpm. Complete parts (a) through (f). (a)...
-
Compare and contrast licensing and subcontracting.
-
For each of the following independent cases, find the probability of the process being in control, at which a manager would be indifferent to investigating the source of the variance. Cost of Cost of...
-
Determing Expected Costs of Investigation and No Investigation} The manager of the Delta Company is always indifferent about conducting an investigation when there is a probability of \(80 \%\) that...
-
Assume that an investigation of a process would cost approximately \(\$ 500\). If the process is out-of-control, the cost of correcting the condition would be \(\$ 1,000\). The present value of cost...

Study smarter with the SolutionInn App

