9. Chemical properties of carbonyl compounds. Chemical properties of mono- and polysaccharides. Theoretical background. Carbonyl compounds...
Fantastic news! We've Found the answer you've been seeking!
Question:






Transcribed Image Text:
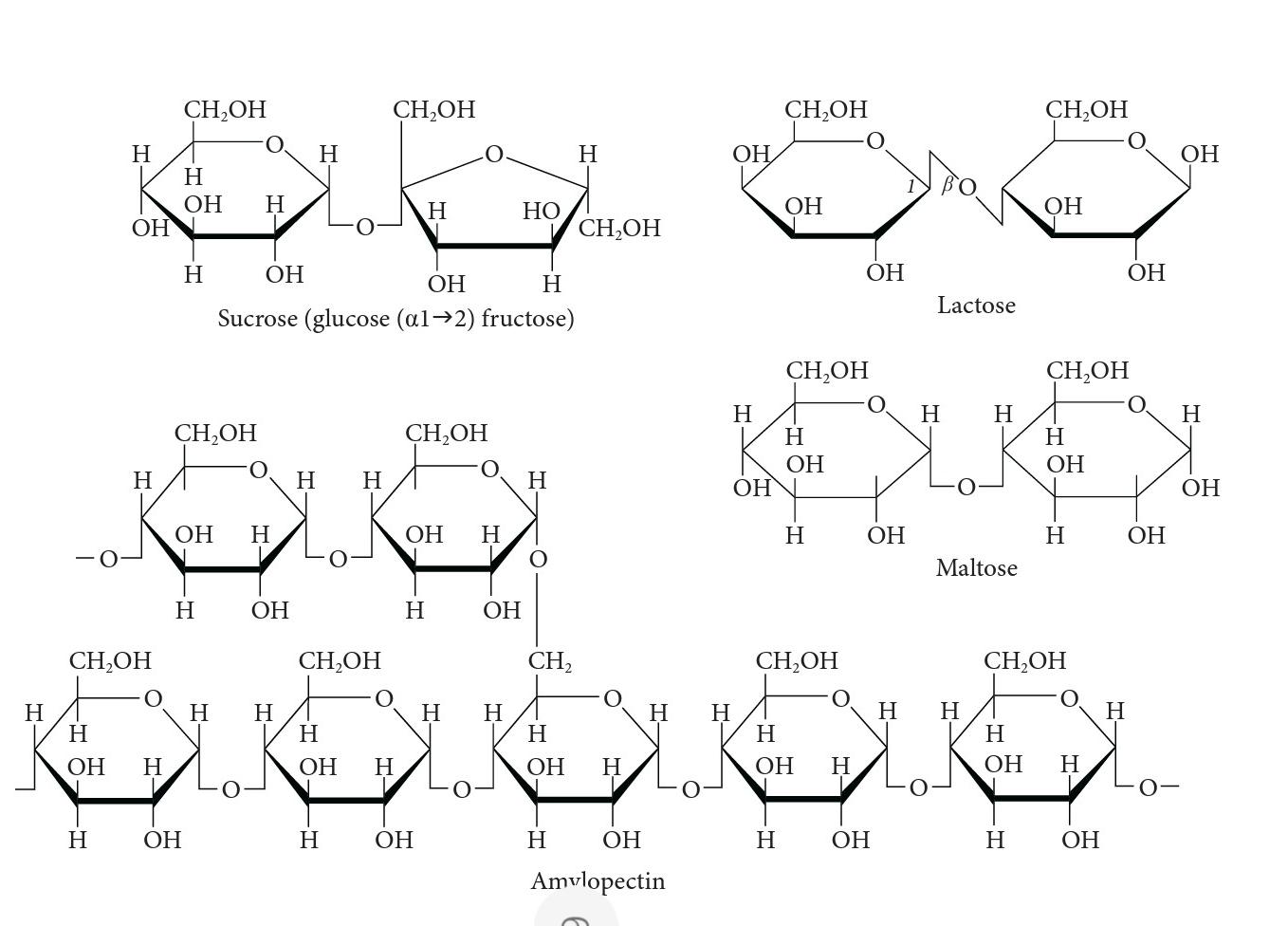
9. Chemical properties of carbonyl compounds. Chemical properties of mono- and polysaccharides. Theoretical background. Carbonyl compounds are organic compounds containing carbonyl (oxo) group in a molecule. The compounds fall into two big groups - aldehydes and ketones. In aldehydes, the carbonyl group has both hydrogen atom and hydrocarbon radical attached. In ketones, the group is bound with two hydrocarbon radicals. According to hydrocarbon radical present, aldehydes are grouped into aliphatic (a), alicyclic (b), and aromatic (c): a) H₂C-C O H Ethanal Cyclohexanecarbaldehyde Benzaldehyde Ketones are also grouped into aliphatic (a), alicyclic, and aromatic. H3C-C-CH3 O (a) Propanone (acetone) H Aldehydes and ketones with up to 4 C atoms in molecules are volatile liquids of specific odour. They are soluble in water and in organic solvents. Solubility in water decreases with an increase of a number of C atoms in a chain. Aldehydes containing 8-10 C atoms in a chain have odour of flowers and are used in perfumery. H₂C-C-CH₂ || O H Properties of carbonyl compounds are determined by chemical prop- erties of both the carbonyl group and hydrocarbon radical. H₂ Propanone (acetone) H₂C-CH-CH₂ I OH 1. Reduction producing alcohols (both aldehydes and ketones): 2. Oxidation producing acids (only aldehydes): In this laboratory work you will prove chemical properties of carbonyl compounds. Experimental part: A. Oxidation of aldehydes Free aldehyde groups can be oxidized to carboxyl easily by mild oxidizing agents - for example, Cu²+ in alkaline solution: R-CHO +2Cu²+ + 5OH- + heating → R-COO + Cu₂O + 3H₂O Red precipitate Blue solution 1. Trommer's reagent: mix of CuSO4 and NaOH; Fehling's reagent: copper is coordinated with sodium potassium tartrate (Fehling-I - CuSO4, Fehling-II - basic sodium potassium tartrate) Procedure 1: Oxidation by Trommer's reagent: Take 2 test tubes and add 6 drops of 2 mol/L NaOH, 6 drops of H₂O and 2 drops of 0.1 mol/L CuSO4 solution into each tube. The blue Cu(OH)₂ precipitate should form in them. 2. 66 Add a few drops of formalin (the solution of formaldehyde) to the 1st tube and a few drops of acetone to the 2nd tube and shake. Heat the test tubes in the heating block until the reaction mixtures change color from blue (Cu²+) to yellow (CuOH) or red (Cu₂O). 4. Describe and explain the observed differences. 3. O || H-C-H →→→H-C-OH Formaldehyde Formic acid O || 1. Procedure 2: Oxidation by Fehling's reagent: Take 2 test tubes; put 5 drops of formalin into the 1st tube and acetone into the 2nd tube. 2. 3. 4. Add 10 drops of Fehling-I and 10 drops of Fehling-II reagents into each tube. Heat the test tubes with solutions in the heating block until the colors of reduced copper compounds appear. Describe and explain the observed differences. B. Reaction of acetone with nitroprusside Sodium nitroprusside is a coordination compound Na₂ [Fe(CN),NO] which forms with acetone a complex compound of intense red color: Procedure: Add 1 drop of 0.25 mol/L sodium nitroprusside solution, 5 drops of water and 1 drop of acetone in the test tube. Add 1 drop of 2 mol/L NaOH - solution becomes red. Add 1 drop of 2 mol/L CH3COOH - the color becomes more intense - almost cherry. 1. CH3-CO-CH3 + Na₂ [Fe(CN)5NO] + 2NaOH → → Na4 [Fe(CN),NO=CH-CO-CH3] + 2H₂O (Intense red) 2. Chemical properties of mono- and polysaccharides Theoretical background. Chemically, carbohydrates are polyhydroxy al- dehydes or polyhydroxy ketones, or substances that yield these compounds when hydrolyzed. Carbohydrates are also known as saccharides. Carbohy- drates are classified as monosaccharides, disaccharides, oligosaccharides and polysaccharides, according to the number of monosaccharide units in a molecule. Monosaccharides usually exist in a cyclic hemiacetal form. The formation of hemiacetal occurs when the oxo group of a monosaccharide reacts with the hydroxyl group of the same monosaccharide. The hemiac- etals of monosaccharaides can react with one another and form disaccharides and polysaccharides. However, hemiacetals are in equilibrium with the small amounts of open chain aldehyde. Because of this, they can be oxidized. Below are the formulas of carbohydrates used in the laboratory work: H 1 C=O T H-C-OH T HO-C-H T H-C-OH T H-C-OH CH₂OH D-Glucose CH₂OH T C=O HO-C-H T H-C-OH T H-C-OH T CH₂OH D-Fructose H OH H H OH CH₂OH H H CH₂OH H OH H H OH OH CH₂OH H ن H OH OH H Sucrose (glucose (al→2) fructose) OH H H H H H CH₂OH H OH H CH2OH H H CH,OH OH H H OH OH HO H H H CH, H H CH₂OH H OH H H OH Amvlopectin H OH H OH CH₂OH ОН H CH₂OH H OH H CH₂OH OH H OH 0. ال OH OH Lactose H H Maltose H H CH₂OH OH H CH₂OH H OH H CH₂OH H OH H OH OH H OH OH H OH Experimental part: C. Reaction of hydroxyl groups of carbohydrates. Adjacent -OH groups present in saccharides form a soluble colored coordination compound with Cu (II) in alkaline solution: T CH-OH CH CH-OH 21 + Cu(OH)₂ + 2NaOH → blue colour precipitate HC-O O-CH T HC-0/ O-CH Cu 12- 2Na+ + 4H₂O water soluble blue complex compound Procedure: 1. Prepare 4 test tubes. 2. Add 3 drops of four different 1% carbohydrate solutions into four separate test tubes: 1st - glucose, 2nd - fructose, 3rd - sucrose and 4th lactose or Trommer's reagent: Fehling's reagent: maltose. 3. Add 6 drops of 2 M NaOH and 2 drop of 0.1 M CuSO4 solution into each tube. The blue Cu(OH)₂ precipitate should form in all the test tubes. After some shaking the precipitate disappears as the blue-colored coordination com- pound between the copper and saccharide is formed. Leave the tubes for the use in following experiments! D. Oxidation of the aldehyde group of carbohydrates. Aldoses exist pri- marily in cyclic hemiacetal form; however, they are in equilibrium with small amounts of open chain aldehyde. Free aldehyde groups can be easily oxidized to carboxyl by mild oxidizing agents - for example, Cu²+ in alka- line solution: R-CHO + 2Cu²+ + 5OH- + heating → R-COO + Cu₂O + 3H₂O Blue solution Red precipitate CuSO4 and NaOH. CuSO4 and basic sodium potassium tartrate. Procedure 1: Oxidation by Trommer's reagent: 1. Add 5 drops of water to each test tube from the previous experiment. 2. Heat the test tubes in the boiling water bath until the reaction mixture changes color from blue (Cu²+) to yellow (CuOH) or red (Cu₂O). 3. Explain the change of color. Procedure 2: Oxidation by Fehling's reagent: 1. Take 4 test tubes, put 5 drops of the same carbohydrates as in the C expe- riment. 2. Add 5 drops of Fehling-I and 5 drops of Fehling-II reagents into each tube. 3. Heat the test tubes in the boiling water bath until the colors of reduced copper compounds appear. E. Hydrolysis of polysaccharides Polysaccharides are cleaved gradually: first into oligosaccharides, then into monosaccharides. This degradation is catalyzed by the specialized en- zymes in vivo, or by acids in vitro. It could be represented by a scheme: Starch (C6H₁0O5) → Soluble starch (C6H1005)n' (where n'<n) →>> Blue violet Violet → Dextrins (C6H1005)n"(n²<n<n) Brown-red Maltose (C₁₂H₂20₁1)→ Glucose (C6H₁2O6) Yellow Yellow The colors given in the scheme are observed when the hydrolysis products react with iodine (1₂). Procedure: Hydrolysis when catalyst is Ht: 1. Take two test tubes. Add 10 drops of 0.5% starch solution into the 1st test tube and add 2 drops of 1 mol/L H₂SO4. 2. The 2nd tube will be the control: put 10 drops of 0.5% starch solution and add 2 drops of water instead of acid. 70 3. Heat both test tubes for approx. 7 min. in the boiling water bath. 4. Dilute iodine solution in another test tube: add 5 drop of iodine solution into 5 mL of water. 5. Put 1 drop of the diluted iodine solution into the hydrolyzed starch solu- tion. Record the observed color. 6. Repeat the same with the control (2nd test tube contents). Compare both colors. Try to estimate which stage of starch hydrolysis was achieved in your experiment. General questions 1. What compounds are called "carbonyl compounds"? What groups of carbonyl compounds do you know? 2. What chemical properties do carbonyl compounds have? 3. Why aldehydes can react with copper sulfate and ketones cannot? What is the principle of a chemical reaction between aldehyde and cop- per sulfate (CuSO4)? 4. Draw an equation of the reaction between formaldehyde and CuSO4. Explain, why the color of the solution changes. What two separate solutions make Fehling's reagent? 5. 6. 7. 8. 9. How are carbohydrates classified? 10. What chemical structures do monosaccharides usually form? 11. What chemical reaction could be performed to prove the existence of What type of reaction occurs when acetone reacts with Na₂ [Fe(CN),NO]? What substances are called "carbohydrates"? hydroxyl groups in carbohydrates? 12. What oxidizers are usually used to oxidize carbohydrates? 13. What substances can catalyze hydrolysis of polysaccharides? 14. What enzyme catalyzes hydrolysis of polysaccharides? 15. What reagents are used to prove hydrolysis of polysaccharides? 7 9. Chemical properties of carbonyl compounds. Chemical properties of mono- and polysaccharides. Theoretical background. Carbonyl compounds are organic compounds containing carbonyl (oxo) group in a molecule. The compounds fall into two big groups - aldehydes and ketones. In aldehydes, the carbonyl group has both hydrogen atom and hydrocarbon radical attached. In ketones, the group is bound with two hydrocarbon radicals. According to hydrocarbon radical present, aldehydes are grouped into aliphatic (a), alicyclic (b), and aromatic (c): a) H₂C-C O H Ethanal Cyclohexanecarbaldehyde Benzaldehyde Ketones are also grouped into aliphatic (a), alicyclic, and aromatic. H3C-C-CH3 O (a) Propanone (acetone) H Aldehydes and ketones with up to 4 C atoms in molecules are volatile liquids of specific odour. They are soluble in water and in organic solvents. Solubility in water decreases with an increase of a number of C atoms in a chain. Aldehydes containing 8-10 C atoms in a chain have odour of flowers and are used in perfumery. H₂C-C-CH₂ || O H Properties of carbonyl compounds are determined by chemical prop- erties of both the carbonyl group and hydrocarbon radical. H₂ Propanone (acetone) H₂C-CH-CH₂ I OH 1. Reduction producing alcohols (both aldehydes and ketones): 2. Oxidation producing acids (only aldehydes): In this laboratory work you will prove chemical properties of carbonyl compounds. Experimental part: A. Oxidation of aldehydes Free aldehyde groups can be oxidized to carboxyl easily by mild oxidizing agents - for example, Cu²+ in alkaline solution: R-CHO +2Cu²+ + 5OH- + heating → R-COO + Cu₂O + 3H₂O Red precipitate Blue solution 1. Trommer's reagent: mix of CuSO4 and NaOH; Fehling's reagent: copper is coordinated with sodium potassium tartrate (Fehling-I - CuSO4, Fehling-II - basic sodium potassium tartrate) Procedure 1: Oxidation by Trommer's reagent: Take 2 test tubes and add 6 drops of 2 mol/L NaOH, 6 drops of H₂O and 2 drops of 0.1 mol/L CuSO4 solution into each tube. The blue Cu(OH)₂ precipitate should form in them. 2. 66 Add a few drops of formalin (the solution of formaldehyde) to the 1st tube and a few drops of acetone to the 2nd tube and shake. Heat the test tubes in the heating block until the reaction mixtures change color from blue (Cu²+) to yellow (CuOH) or red (Cu₂O). 4. Describe and explain the observed differences. 3. O || H-C-H →→→H-C-OH Formaldehyde Formic acid O || 1. Procedure 2: Oxidation by Fehling's reagent: Take 2 test tubes; put 5 drops of formalin into the 1st tube and acetone into the 2nd tube. 2. 3. 4. Add 10 drops of Fehling-I and 10 drops of Fehling-II reagents into each tube. Heat the test tubes with solutions in the heating block until the colors of reduced copper compounds appear. Describe and explain the observed differences. B. Reaction of acetone with nitroprusside Sodium nitroprusside is a coordination compound Na₂ [Fe(CN),NO] which forms with acetone a complex compound of intense red color: Procedure: Add 1 drop of 0.25 mol/L sodium nitroprusside solution, 5 drops of water and 1 drop of acetone in the test tube. Add 1 drop of 2 mol/L NaOH - solution becomes red. Add 1 drop of 2 mol/L CH3COOH - the color becomes more intense - almost cherry. 1. CH3-CO-CH3 + Na₂ [Fe(CN)5NO] + 2NaOH → → Na4 [Fe(CN),NO=CH-CO-CH3] + 2H₂O (Intense red) 2. Chemical properties of mono- and polysaccharides Theoretical background. Chemically, carbohydrates are polyhydroxy al- dehydes or polyhydroxy ketones, or substances that yield these compounds when hydrolyzed. Carbohydrates are also known as saccharides. Carbohy- drates are classified as monosaccharides, disaccharides, oligosaccharides and polysaccharides, according to the number of monosaccharide units in a molecule. Monosaccharides usually exist in a cyclic hemiacetal form. The formation of hemiacetal occurs when the oxo group of a monosaccharide reacts with the hydroxyl group of the same monosaccharide. The hemiac- etals of monosaccharaides can react with one another and form disaccharides and polysaccharides. However, hemiacetals are in equilibrium with the small amounts of open chain aldehyde. Because of this, they can be oxidized. Below are the formulas of carbohydrates used in the laboratory work: H 1 C=O T H-C-OH T HO-C-H T H-C-OH T H-C-OH CH₂OH D-Glucose CH₂OH T C=O HO-C-H T H-C-OH T H-C-OH T CH₂OH D-Fructose H OH H H OH CH₂OH H H CH₂OH H OH H H OH OH CH₂OH H ن H OH OH H Sucrose (glucose (al→2) fructose) OH H H H H H CH₂OH H OH H CH2OH H H CH,OH OH H H OH OH HO H H H CH, H H CH₂OH H OH H H OH Amvlopectin H OH H OH CH₂OH ОН H CH₂OH H OH H CH₂OH OH H OH 0. ال OH OH Lactose H H Maltose H H CH₂OH OH H CH₂OH H OH H CH₂OH H OH H OH OH H OH OH H OH Experimental part: C. Reaction of hydroxyl groups of carbohydrates. Adjacent -OH groups present in saccharides form a soluble colored coordination compound with Cu (II) in alkaline solution: T CH-OH CH CH-OH 21 + Cu(OH)₂ + 2NaOH → blue colour precipitate HC-O O-CH T HC-0/ O-CH Cu 12- 2Na+ + 4H₂O water soluble blue complex compound Procedure: 1. Prepare 4 test tubes. 2. Add 3 drops of four different 1% carbohydrate solutions into four separate test tubes: 1st - glucose, 2nd - fructose, 3rd - sucrose and 4th lactose or Trommer's reagent: Fehling's reagent: maltose. 3. Add 6 drops of 2 M NaOH and 2 drop of 0.1 M CuSO4 solution into each tube. The blue Cu(OH)₂ precipitate should form in all the test tubes. After some shaking the precipitate disappears as the blue-colored coordination com- pound between the copper and saccharide is formed. Leave the tubes for the use in following experiments! D. Oxidation of the aldehyde group of carbohydrates. Aldoses exist pri- marily in cyclic hemiacetal form; however, they are in equilibrium with small amounts of open chain aldehyde. Free aldehyde groups can be easily oxidized to carboxyl by mild oxidizing agents - for example, Cu²+ in alka- line solution: R-CHO + 2Cu²+ + 5OH- + heating → R-COO + Cu₂O + 3H₂O Blue solution Red precipitate CuSO4 and NaOH. CuSO4 and basic sodium potassium tartrate. Procedure 1: Oxidation by Trommer's reagent: 1. Add 5 drops of water to each test tube from the previous experiment. 2. Heat the test tubes in the boiling water bath until the reaction mixture changes color from blue (Cu²+) to yellow (CuOH) or red (Cu₂O). 3. Explain the change of color. Procedure 2: Oxidation by Fehling's reagent: 1. Take 4 test tubes, put 5 drops of the same carbohydrates as in the C expe- riment. 2. Add 5 drops of Fehling-I and 5 drops of Fehling-II reagents into each tube. 3. Heat the test tubes in the boiling water bath until the colors of reduced copper compounds appear. E. Hydrolysis of polysaccharides Polysaccharides are cleaved gradually: first into oligosaccharides, then into monosaccharides. This degradation is catalyzed by the specialized en- zymes in vivo, or by acids in vitro. It could be represented by a scheme: Starch (C6H₁0O5) → Soluble starch (C6H1005)n' (where n'<n) →>> Blue violet Violet → Dextrins (C6H1005)n"(n²<n<n) Brown-red Maltose (C₁₂H₂20₁1)→ Glucose (C6H₁2O6) Yellow Yellow The colors given in the scheme are observed when the hydrolysis products react with iodine (1₂). Procedure: Hydrolysis when catalyst is Ht: 1. Take two test tubes. Add 10 drops of 0.5% starch solution into the 1st test tube and add 2 drops of 1 mol/L H₂SO4. 2. The 2nd tube will be the control: put 10 drops of 0.5% starch solution and add 2 drops of water instead of acid. 70 3. Heat both test tubes for approx. 7 min. in the boiling water bath. 4. Dilute iodine solution in another test tube: add 5 drop of iodine solution into 5 mL of water. 5. Put 1 drop of the diluted iodine solution into the hydrolyzed starch solu- tion. Record the observed color. 6. Repeat the same with the control (2nd test tube contents). Compare both colors. Try to estimate which stage of starch hydrolysis was achieved in your experiment. General questions 1. What compounds are called "carbonyl compounds"? What groups of carbonyl compounds do you know? 2. What chemical properties do carbonyl compounds have? 3. Why aldehydes can react with copper sulfate and ketones cannot? What is the principle of a chemical reaction between aldehyde and cop- per sulfate (CuSO4)? 4. Draw an equation of the reaction between formaldehyde and CuSO4. Explain, why the color of the solution changes. What two separate solutions make Fehling's reagent? 5. 6. 7. 8. 9. How are carbohydrates classified? 10. What chemical structures do monosaccharides usually form? 11. What chemical reaction could be performed to prove the existence of What type of reaction occurs when acetone reacts with Na₂ [Fe(CN),NO]? What substances are called "carbohydrates"? hydroxyl groups in carbohydrates? 12. What oxidizers are usually used to oxidize carbohydrates? 13. What substances can catalyze hydrolysis of polysaccharides? 14. What enzyme catalyzes hydrolysis of polysaccharides? 15. What reagents are used to prove hydrolysis of polysaccharides? 7
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Compounds that contain both carbonyl and alcohol functional groups are often more stable as cyclic hemiacetals or cyclic acetals than as open-chain compounds. Examples of several of these are shown....
-
Organic compounds containing radioactive atoms can be used to follow the biosynthesis of molecules in cells and organisms. In these experiments, the amount of radioisotope in proteins derived from...
-
Volatile organic compounds (VOCs) can be removed from water effluents by stripping in packed towers. Possible stripping agents are steam and air. Alternatively, the VOCs can be removed by carbon...
-
15. Show that the frequency of revolution of the electron in the Bohr model hydrogen atom is given by v = 2|E|/hn where E is the total energy of the electron. 16. Show that for all Bohr orbits the...
-
Presented below is financial information related to the 2014 operations of Donna Marie Cruise Company. Maintenance and repairs expense ........$ 97,000 Utilities expense ................ 10,000...
-
List and describe the three approaches to policy development presented in this chapter. In your opinion, which is best suited for use by a smaller organization and why? If the target organization...
-
Determine the missing amounts in the following accounting equations. 1. Label each of the transactions in the preceding analysis with the correspon ding letter of the description that best fits it:...
-
Answer the following questions. a. Which department is responsible for initiating the purchase of materials? b. What is the name of the document generated by the department identified in (a) above?...
-
Please solve correctly C C D Question 11 10 Points 1. As an entrepreneur, you must be aware of your stakeholders and business ethics and define the obligation to each one of them to your business...
-
Find the risk-neutral probabilities and zero cost date 0 forward prices (for settlement at date 1) for the stock in exercise 7.2. As in that exercise, assume a risk-free rate of 15% per period. 7.2....
-
The company: TICKER EMP# Market Capitilization BETA GREENIDGE GENERATION HOLDINGS INC. XNAS:GREE 347 32058390 3.623 As a CPA, you have received an offer to...
-
Discuss the potential impact this could have on future legislation. For instance, do you feel the democratic party represents the voice of the people?
-
Puan Rosmah, a retired lecturer plans to invest some money in buying common shares. She expects a return of 12% from all her investments. She was given the following information on the stocks of...
-
Part 1) Consider the Cobb-Douglas production function: Y = AK-aLa, where K and L are the capital stock and labor uses respectively. Consider that the economy is composed of a "representative" firm...
-
In regard to section 32 of the Sale of Land Act 1962 (VIC), which part of it will apply for a scenario when a purchaser found out the property he's bought and awaiting settlement is smaller than...
-
Agnes is a speculator in investing in options. Agnes had purchased a put option on Australian dollar with a strike price of $0.80. the premium is $0.02. on the expiration date, the Australian dollar...
-
A 10%, 10 years bond of Tk. 5,000 has been issued at 10% discount and redeemed at 5% premium. If flotation cost is 2%., what is the approximate YTM
-
What are the principal alloying elements in SAE 4340 steel?
-
Would you expect each of the following atoms to gain or lose electrons when forming ions? What ion is the most likely in each case? a. Ra b. In c. P d. Te e. Br f. Rb
-
A spherical glass container of unknown volume contains helium gas at 25 o C and 1.960 atm. When a portion of the helium is withdrawn and adjusted to 1.00 atm at 25 o C, it is found to have a volume...
-
Consider the following pH curves for 100.0 mL of two different acids with the same intital concentration each titrated by 0.10 M NaOH: a. Which plot represents a pH curve of a weak acid, and which...
-
Fill in the Blank. When finite amplitudes of motion are involved, __________ analysis becomes necessary.
-
Fill in the Blank. __________ equation involves time-dependent coefficients.
-
Fill in the Blank. __________ principle is not applicable in nonlinear analysis.
Migrating To The Cloud Oracle Client Server Modernization 1st Edition - ISBN: 1597496472 - Free Book

Study smarter with the SolutionInn App


