A 100 kmol of natural gas is burned with 20% of excess pure O. The natural...
Fantastic news! We've Found the answer you've been seeking!
Question:
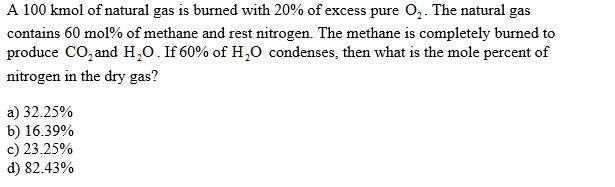
Transcribed Image Text:
A 100 kmol of natural gas is burned with 20% of excess pure O₂. The natural gas contains 60 mol% of methane and rest nitrogen. The methane is completely burned to produce CO₂ and H₂O. If 60% of H₂O condenses, then what is the mole percent of nitrogen in the dry gas? a) 32.25% b) 16.39% c) 23.25% d) 82.43% A 100 kmol of natural gas is burned with 20% of excess pure O₂. The natural gas contains 60 mol% of methane and rest nitrogen. The methane is completely burned to produce CO₂ and H₂O. If 60% of H₂O condenses, then what is the mole percent of nitrogen in the dry gas? a) 32.25% b) 16.39% c) 23.25% d) 82.43%
Expert Answer:
Answer rating: 100% (QA)
Answer Solution sine CH4 20 Metham reaction combustion reaction cot 2420 Took... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these finance questions
-
Natural gas is burned with air to produce gaseous products at 1985C. Express this temperature in K, R and F.
-
Ethane (C 2 H 6 ) is burned with 20 percent excess air during a combustion process. Assuming complete combustion and a total pressure of 100 kPa, determine (a) The airfuel ratio (b) The dew-point...
-
A sample of natural gas is 85.2% methane, CH4, and 14.8% ethane, C2H6, by mass. What is density of this mixture at 18oC and 771 mmHg?
-
(CMA, adapted) Roletter Company makes and sells artistic frames for pictures of weddings, graduations, and other special events. Bob Anderson, the controller, is responsible for preparing Roletters...
-
Dean Inc. has negotiated the purchase of a new piece of automatic equipment at a price of $16,000 plus trade-in, f.o.b. factory. Dean Inc. paid $16,000 cash and traded in used equipment. The used...
-
Santana Rey receives the March bank statement for Business Solutions on April 11, 2018. The March 31 bank statement shows an ending cash balance of $67,566. A comparison of the bank statement with...
-
The FBI computed the proportion of violent crimes in the United States falling into each of four categories. A simple random sample of 500 violent crimes committed in California were categorized in...
-
Patton issues $650,000 of 5%, four-year bonds dated January 1, 2011, that pay interest semiannually on June 30 and December 31. They are issued at $584,361 and their market rate is 8% at the issue...
-
A public sector organization is creating budgets to assess the performance of all its departments. One of those departments is dealing with elderly citizens' well-being and is named...
-
Wayland Custom Woodworking is a firm that manufactures custom cabinets and woodwork for business and residential customers. Students will have the opportunity to establish payroll records and to...
-
Three moles of an ideal gas (with temperature-independent Cp =(7/2)R, Cv = (5/2)R) is contained in a horizontal piston/cylinderarrangement. The piston has an area of 0.1m^2 and mass of 500 g.The...
-
Write an original summary of the article on how families can contribute negatively to college students' success outcomes. Summaries should not include direct quotations, instead use your own words to...
-
Ms. Jones is a forty-seven-year old female, referred for examination after she had partially disemboweled a young man, apparently without any provocation. Ms. Jones occupied an apartment on the...
-
How much and in what ways has the launch of the Cayenne changed the meaning of the Porsche brand? What is the value of the Rennlist online brand community to Porsche? How does it help and/or hurt the...
-
Let 3i4j; find |||| =
-
Bailey Delivery Company, Incorporated, was organized in 2021 in Wisconsin. The following transactions occurred during the year: a. Received cash from investors in exchange for 17,000 shares of stock...
-
When two parties have bottom lines that permit an agreement at some point between them, there is a _____ bargaining zone. When the two bottom lines do not overlap, there is a _____ bargaining zone....
-
Which, if any, of the dichloroethene molecules drawn in Data Table II (3.) (4.) and (5.) are geometric isomers? A. B. C. D. cis-1,2-dichloroethene and trans-1,2-dichloroethene...
-
Why does a nonquasi-equilibrium compression process require a larger work input than the corresponding quasi-equilibrium one?
-
A solar cell has an open circuit voltage value of 0.60 V with a reverse saturation current density of Jo = 3.9 109 A/m2. The temperature of the cell is 27oC, the cell voltage is 0.52 V, and the cell...
-
An adiabatic heat exchanger is to cool ethylene glycol (cp = 2.56 kJ/kg C) flowing at a rate of 2 kg/s from 80 to 40C by water (cp = 4.18 kJ/kg C) that enters at 20C and leaves at 55C. Determine...
-
Fill in the Blank. In the finite element method, the solution domain is replaced by several ___________.
-
The simplest form of mass matrix is known as a. lumped-mass matrix b. consistent-mass matrix c. global mass matrix
-
What is a shape function?

Study smarter with the SolutionInn App


