A 35% solution of Na2CO3 weighing 6000 kg is cooled to 20C to yield crystals of...
Fantastic news! We've Found the answer you've been seeking!
Question:
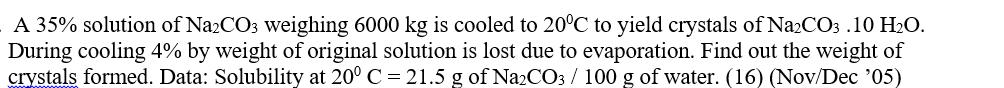
Transcribed Image Text:
A 35% solution of Na2CO3 weighing 6000 kg is cooled to 20°C to yield crystals of Na2CO3.10 H₂O. During cooling 4% by weight of original solution is lost due to evaporation. Find out the weight of crystals formed. Data: Solubility at 20° C = 21.5 g of Na2CO3 / 100 g of water. (16) (Nov/Dec '05) A 35% solution of Na2CO3 weighing 6000 kg is cooled to 20°C to yield crystals of Na2CO3.10 H₂O. During cooling 4% by weight of original solution is lost due to evaporation. Find out the weight of crystals formed. Data: Solubility at 20° C = 21.5 g of Na2CO3 / 100 g of water. (16) (Nov/Dec '05)
Expert Answer:
Answer rating: 100% (QA)
We can start by using the information provided to find out the weight of water evaporated ... View the full answer

Related Book For 

Materials Science and Engineering An Introduction
ISBN: 978-1118324578
9th edition
Authors: William D. Callister Jr., David G. Rethwisch
Posted Date:
Students also viewed these chemical engineering questions
-
The solubility of Cl2 in 100 g of water at STP is 310 cm3. Assume that this quantity of Cl2 is dissolved and equilibrated as follows: (a) If the equilibrium constant for this reaction is 4.7 Ã...
-
A uniform plate girder weighing 6000 lb is held in a horizontal position by two crane cables. Determine the angle α and the tension in each cable. 30 6 ft 20 ft 60 ft
-
The solubility of carbon dioxide in water is 0.161 g CO2 in 100 mL of water at 20C and 1.00 atm. A soft drink is carbonated with carbon dioxide gas at 5.50 atm pressure. What is the solubility of...
-
Let F = [F] lb. a) Determine the force Fp acting at roller D. b) Determine the force FE acting at pin E. F 4 ft B 600 lb. ft -3 ft C -3 ft OD -2 ft- E
-
How do centralized and decentralized firms differ? What are the advantages of each?
-
Who is ultimately responsible for identifying new or emerging key risk areas that should be covered by the organizations governance process? a. The board of directors. b. Senior management. c. Risk...
-
A \(2.0-\mathrm{H}\) inductor carries a current that is increasing at a rate of \(0.40 \mathrm{~A} / \mathrm{s}\). What is the magnitude of the \(\mathrm{emf}\) induced in the inductor? Does this...
-
Timothy Monroe opened a law office on January 1, 2017. During the first month of operations, the business completed the following transactions: Jan. 1 Monroe contributed $62,000 cash to the business,...
-
Kericho Sports Club has been operating for some time now. The treasurer analysed the cash book and produced the following Receipts and Payments account for the year ended 31st Dec. 2018. Receipts...
-
Liquid reaction A --> 2B is taking place in a steady state packed bed reactor that has total available packing area (A) of 10 m. The density (p) and packing surface (a) of the catalyst per volume are...
-
Approximately 14% of the population of Honduras lives in extreme poverty (defined as less than $2.15 per day at 2017 PPP). Imagine that the government of Honduras partners with the Inter-American...
-
Your firm has a risk-free investment opportunity where it can invest $160,000 today and receive $170,000 in one year. For what level of interest rates is this project attractive?
-
In the late 1990s, the city of New York confronted growing concerns surrounding the mosquito-borne West Nile virus by implementing an aerial and ground spraying program. The program consisted of...
-
Dayenian entered into a two-year lease agreement for an apartment with Monticello Realty Corporation on July 14. On October 26 of that year, she signed a portion of the lease headed Assignment, which...
-
Search the Internet for four accounts of successful corporate entrepreneurship. What key factors for success are common across all these accounts? Which are unique? If one company can foster an...
-
Isnt corporate entrepreneurship an oxymoron? Do the characteristics of an established organization, such as its routines and structure, increase efficiency but at the same time kill any...
-
K As the manager of the human resources department at your firm, you are investigating the number of sick days employees took during the last calendar year. For employees in the marketing area, the...
-
From 1970 to 1990, Sri Lanka's population grew by approximately 2.2 million persons every five years. The population in 1970 was 12.2 million people.What is the best formula for P, Sri Lanka's...
-
The fatigue data for a steel alloy are given as follows: Stress Amplitude [MPa (ksi)] __________ Cycles to Failure 470 (68.0) ................................... 104 440 (63.4)...
-
Atomic radius, crystal structure, electro negativity, and the most common valence are given in the following table for several elements; for those that are nonmetals, only atomic radii are indicated....
-
A cylindrical bar of aluminum 19 mm (0.75 in.) in diameter is to be deformed elastically by application of a force along the bar axis. Using the data in Table 6.1, determine the force that produces...
-
Suppose you operate a bank in a country where the central bank is expected to embark on a series of interest rate increases. Based on gap analysis, would this scenario be more likely to hurt or help...
-
Cyber risk has been recognized as a growing source of operational risk for financial institutions. Why might managing this risk at an individual firm level not be adequate?
-
Bank assets equal bank liabilities plus bank capital. a. Bank assets are the uses for bank funds. i. They include reserves, securities, and loans. ii. Over the years, commercial and industrial loans...

Study smarter with the SolutionInn App


