A 64.3 mg sample of a protein (MW = 58,600) was treated with 2.00 mL of...
Fantastic news! We've Found the answer you've been seeking!
Question:
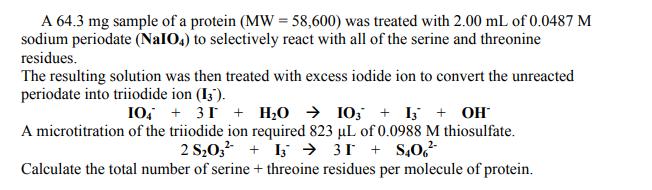
Transcribed Image Text:
A 64.3 mg sample of a protein (MW = 58,600) was treated with 2.00 mL of 0.0487 M sodium periodate (NaIO4) to selectively react with all of the serine and threonine residues. The resulting solution was then treated with excess iodide ion to convert the unreacted periodate into triiodide ion (13). 10 +31 + H₂O → 103 + 13 + OH A microtitration of the triiodide ion required 823 µL of 0.0988 M thiosulfate. 2 S₂03²1331+S406² Calculate the total number of serine + threoine residues per molecule of protein. A 64.3 mg sample of a protein (MW = 58,600) was treated with 2.00 mL of 0.0487 M sodium periodate (NaIO4) to selectively react with all of the serine and threonine residues. The resulting solution was then treated with excess iodide ion to convert the unreacted periodate into triiodide ion (13). 10 +31 + H₂O → 103 + 13 + OH A microtitration of the triiodide ion required 823 µL of 0.0988 M thiosulfate. 2 S₂03²1331+S406² Calculate the total number of serine + threoine residues per molecule of protein.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A 4.7 10 -2 mg sample of a protein is dissolved in water to make 0.25 mL of solution. The osmotic pressure of the solution is 0.56 torr at 25 C. What is the molar mass of the protein?
-
A 4.7 10 -2 mg sample of a protein is dissolved in water to make 0.25 mL of the solution. The osmotic pressure of the solution is 0.56 torr at 25C. What is the molar mass of the protein?
-
Calculate the sodium ion concentration when 70.0 mL of 3.0 M sodium carbonate is added to 30.0 mL of 1.0 M sodium bicarbonate. Discuss.
-
Two wheels A and B have masses m A and m B and radii of gyration about their central vertical axes of k A and k B respectively. If they are freely rotating in the same direction at A and about the...
-
No-Growth Industries pays out all of its earnings as dividends. It will pay its next $4 per share dividend in a year. The discount rate is 12%. a. What is the price-earnings ratio of the company? b....
-
In Problems 5 16, evaluate each expression. 60 20
-
Describe the working of internal shoe expanding brake with a neat sketch. Also, derive the expression for braking torque.
-
The inventory accounting records for Lee Enterprises contained the following data: Beginning inventory ........ 400 units at $12 each Purchase 1, Feb. 26 .........2,300 units at $14 each Sale, March...
-
A 20N force acts perpendicular to the door 0.8m wide at its edge. Find moment at hinges. Also find moment at hinges if 20N force acts at 60 with the plane of door. i) 20N force acting perpendicular...
-
Tammy Touchtone operates a talent agency called Touchtone Talent Agency. Some clients pay in advance for services; others are billed after services have been performed. Advance payments are credited...
-
During 2017, Standard Bank faced many problems that they needed to solve, management needed to go through several stages that help them think through the problem and make optimal decisions with...
-
The ledger account balances for Greely Corporation at December 31, 2022 are as follows: Cash $ 300 Accounts Receivable 522 Prepaid Insurance 82 Supplies 180 Equipment 4,000 Accumulated Depreciation,...
-
A recipe for baba ghannuj (pured roasted eggplant) requires 1 ounces of pured eggplant for each serving. If you make enough baba ghannuj to serve 300 people, how many eggplants should you purchase?...
-
Completely expand the following logarithmic expressions. Wherever possible, evaluate logarithmic expressions. A) In(yet) B) x6 log5 125 C) 32ab2 log2 de
-
1. (12 points) Consider the following context-free grammar G and answer the following questions. S A1B A 0A | B B1 | (a) What are the variables, terminals, and the start variable of G? (b) For each...
-
Table 1 below shows the sieve size and corresponding weight retained for 3 different stockpiles. Calculate the percent passing for all the 3 aggregate stockpiles. (20 points) Sieve size (mm) 19 13.2...
-
Consider a duopoly with each firm having different marginal costs. Each firm has a marginal cost curve MC = 10 + 2Q; for i = 1, 2. The market demand curve is P = 20 - Q, where Q = Q + Q. 1. What are...
-
KD Insurance Company specializes in term life insurance contracts. Cash collection experience shows that 20 percent of billed premiums are collected in the month before they are due, 60 percent are...
-
Suppose benzene contained three distinct single bonds and three distinct double bonds. How many different isomers would there be for dichlorobenzene (C6H4Cl2)? Draw all your proposed structures.
-
Starting with magnesium and concentrated nitric acid, describe how you would prepare magnesium oxide.
-
A solution is prepared by condensing 4.00 L of a gas, measured at 27C and 748 mmHg pressure, into 58.0 g of benzene. Calculate the freezing point of this solution.
-
Financial balances for the car hire business of Terry's Wedding Cars on 31 March 2024 are provided below in a table in accounting equation form similar to the chapter illustrations. During April, the...
-
Trans Clothing Alterations began operations on 1 August 2024 and completed the following transactions during the first month. 1. Tran deposited \($18\) 000 of her personal funds in a current account...
-
Finesse Fitness was established on 1 April 2024 with an initial investment of $60000 by the owner, Daniel Hewitt. During the first few months of business, the owner employed a student studying...

Study smarter with the SolutionInn App



