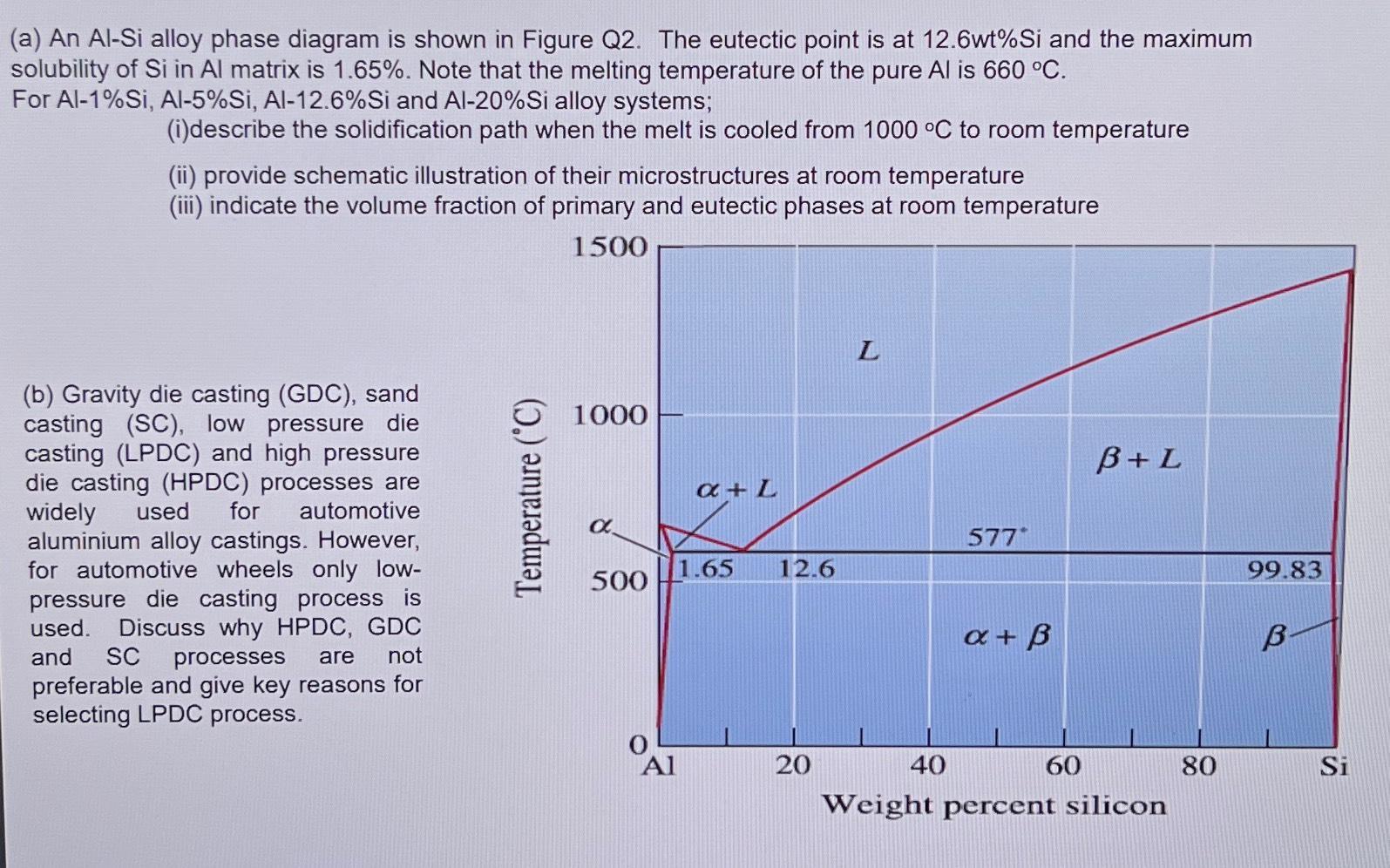
(a) An Al-Si alloy phase diagram is shown in Figure Q2. The eutectic point is at...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
(a) An Al-Si alloy phase diagram is shown in Figure Q2. The eutectic point is at 12.6wt%Si and the maximum solubility of Si in Al matrix is 1.65%. Note that the melting temperature of the pure Al is 660 °C. For Al-1% Si, Al-5%Si, Al-12.6%Si and Al-20%Si alloy systems; (i)describe the solidification path when the melt is cooled from 1000 °C to room temperature (ii) provide schematic illustration of their microstructures at room temperature (iii) indicate the volume fraction of primary and eutectic phases at room temperature 1500 (b) Gravity die casting (GDC), sand casting (SC), low pressure die casting (LPDC) and high pressure die casting (HPDC) processes are widely used for automotive aluminium alloy castings. However, for automotive wheels only low- pressure die casting process is used. Discuss why HPDC, GDC and SC processes are not preferable and give key reasons for selecting LPDC process. Temperature (°C) 1000 500 Al a + L 1.65 12.6 20 L 40 577 x + ß 60 B+L Weight percent silicon 80 99.83 B Si (a) An Al-Si alloy phase diagram is shown in Figure Q2. The eutectic point is at 12.6wt%Si and the maximum solubility of Si in Al matrix is 1.65%. Note that the melting temperature of the pure Al is 660 °C. For Al-1% Si, Al-5%Si, Al-12.6%Si and Al-20%Si alloy systems; (i)describe the solidification path when the melt is cooled from 1000 °C to room temperature (ii) provide schematic illustration of their microstructures at room temperature (iii) indicate the volume fraction of primary and eutectic phases at room temperature 1500 (b) Gravity die casting (GDC), sand casting (SC), low pressure die casting (LPDC) and high pressure die casting (HPDC) processes are widely used for automotive aluminium alloy castings. However, for automotive wheels only low- pressure die casting process is used. Discuss why HPDC, GDC and SC processes are not preferable and give key reasons for selecting LPDC process. Temperature (°C) 1000 500 Al a + L 1.65 12.6 20 L 40 577 x + ß 60 B+L Weight percent silicon 80 99.83 B Si
Expert Answer:
Answer rating: 100% (QA)
a i Solidification Path Al1Si alloy The alloy will solidify as a singlephase solid solution of aluminum with dispersed silicon atoms Al5Si alloy Upon cooling primary silicon will start to form first f... View the full answer

Related Book For 

Essentials of Materials Science and Engineering
ISBN: 978-1111576851
3rd edition
Authors: Donald R. Askeland, Wendelin J. Wright
Posted Date:
Students also viewed these accounting questions
-
The copper-silver phase diagram is shown in Figure 11-30. Copper has a higher melting point than silver. Refer to the silver-rich solid phase as gamma (γ) and the copper-rich solid phase...
-
Read the Speech of Martin Luther King, Jr.'s "I Have a Dream" and answer the question: Kin, makes liberal use of metaphorand metaphorical imagery in his speech. (Glossary: Figures of Speech) Choose a...
-
You are heading toward an island in your speedboat when you see a friend standing on shore at the base of a cliff. You sound the boat's horn to get your friend's attention. Let the wavelength of the...
-
The output at a certain factory is Q(L) = 600L 2/3 units, where L is the size of the labor force. The manufacturer wishes to increase output by 1%. Use calculus to estimate the percentage increase in...
-
Air flows steadily between two cross sections in a long, straight section of \(0.1-\mathrm{m}\)-inside-diameter pipe. The static temperature and pressure at each section are indicated in Fig. P5.9....
-
The management team of Magnificent Modems, Inc. (MMI), wants to investigate the effect of several different growth rates on sales and cash receipts. Cash sales for the month of January are expected...
-
Weller Company's budgeted unit sales for next year are provided below: Budgeted unit sales 1st Quarter 2nd Quarter 3rd Quarter 4th Quarter 26,000 27,000 19,000 24,000 The company's variable selling...
-
Garvey Company sells machine parts to industrial equipment manufacturers for an average price of $0.75 per part. There are two types of customers: those who place small, frequent orders and those who...
-
What is the SWOT Analysis of GoTransit Canada. Provide brief explanation for every strength, weakness, opportunity, and threat.
-
Unlike Federal (which has progressive rates), Indiana has a flat income tax rate of what amount?
-
Yellow House is an office building comprising 2,000 square metres Net Lettable Area (NLA). It was let one and a half years ago on Full Repairing and Insuring (FRI) terms at 305,000 per annum for a...
-
What is the net income of sole proprietorships, partnerships and other unincorporated businesses?
-
Find the center of mass of the solid given by W = {(p, 0, 0): 0 p 1, 0 < 0 2, 0
-
Sofia plans to save for next two years. She plans to put away $1000 every 6 months starting 6 months from now. What will be the value of her savings after two years if her savings accounts earns at a...
-
The given histogram shows the frequency distribution of the speed of cars passing through a particular spot on a highway. Read the graph and answer the following question. 100 90 90 80 70 60 50 40 30...
-
Separate variables and use partial fractions to solve the initial value problems in Problems 18. Use either the exact solution or a computer-generated slope field to sketch the graphs of several...
-
Determine the composition range in which the Tl-Pb alloy at 350 C is (a) Fully liquid; (b) Fully solid;
-
We would like to produce a quenched and tempered aluminum bronze containing 13% Al. Recommend a heat treatment, including appropriate temperatures. Calculate the amount of each phase after each step...
-
The diffusion of yttrium ions along grain boundaries in chromium oxide (Cr2CO3) has been studied by Lesage and co-workers. Their data for grain-boundary diffusivities are shown here. (a) From these...
-
See the option quote on IBM from the CBOE Web site on the next page showing options expiring in March and April 2022. a. Which option contract had the most trades that day? b. Which option contract...
-
You are an options dealer who deals in non-publicly traded options. One of your clients wants to purchase a one-year European call option on HAL Computer Systems stock with a strike price of \($20.\)...
-
It is February 21, 2022, and you have decided to purchase 10 June call contracts on eBays stock with an exercise price of \($57.50.\) Because you are buying, you must pay the ask price. How much...

Study smarter with the SolutionInn App


