A. B. C. D. In which of the following reactions will Kc = Kp? 4 NH3(g)...
Fantastic news! We've Found the answer you've been seeking!
Question:
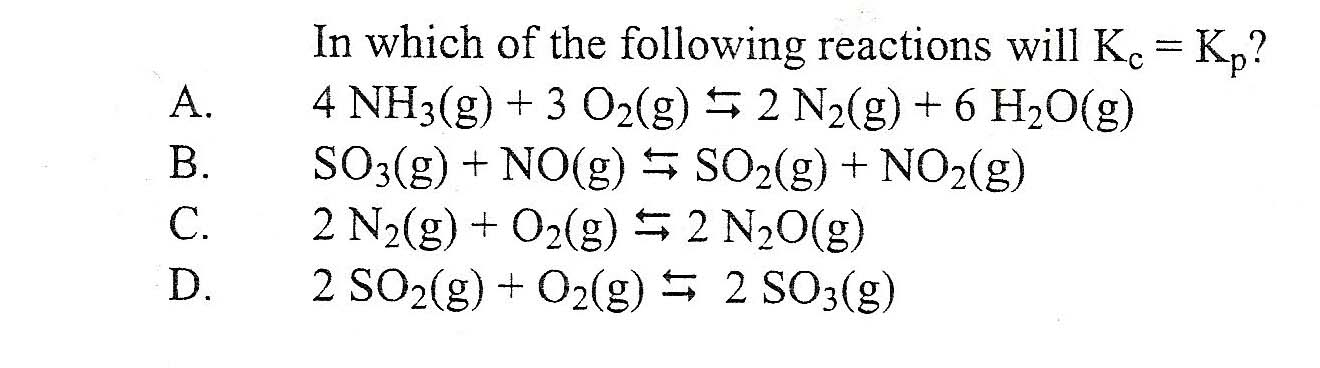
Transcribed Image Text:
A. B. C. D. In which of the following reactions will Kc = Kp? 4 NH3(g) + 3 O₂(g) ≤ 2 N₂(g) + 6 H₂O(g) SO3(g) + NO(g) SO₂(g) + NO₂(g) 2 N₂(g) + O₂(g) ⇒ 2 N₂O(g) 2 SO2(g) + O₂(g) ⇒ 2 SO3(g) A. B. C. D. In which of the following reactions will Kc = Kp? 4 NH3(g) + 3 O₂(g) ≤ 2 N₂(g) + 6 H₂O(g) SO3(g) + NO(g) SO₂(g) + NO₂(g) 2 N₂(g) + O₂(g) ⇒ 2 N₂O(g) 2 SO2(g) + O₂(g) ⇒ 2 SO3(g)
Expert Answer:
Answer rating: 100% (QA)
a 3 when on 0 then kpkc on Sum of no of moles of gaseons Products ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Which of the following reactions occurs most rapidly? Why? a. b. c. Br - H20 C(CH3)3 C(CH3)a (CH)a C(CH3)3 Br - H20 CH33 C(CH3)3
-
Which of the following reactions and decays are possible? For those forbidden, explain what laws are violated. (a) + p n + 0 (b) + + p n + 0 (c) + + p p + e0 (d) p e+ + vp (e) + e+ + vp (f) p n + e...
-
Which of the following reactions are possible, and by what interaction could they occur? For those forbiddevn explain why. (a) + p K0 + p + 0 (b) K + p 0 + 0 (c) K+ + n + + 0 + y (d) K + 0 + 0 + +...
-
Draw a UML Sequence Diagram for the process involved in paying a vendor for an item in cash. The process to follow is below. [15 points] a) Go to the bank b) Request your account balance c) The bank...
-
Develop an OC curve for a sampling plan in which a sample of n = 5 items is drawn from lots of N = 1000 items. The accept/reject criteria are set up in such a way that we accept a lot if no more than...
-
Identify several ways in which you currently use accounting information in your life as a student. Also identify several situations in which, while you are still a student, you might be required to...
-
Repeat the calculations of Example 6.3, but for \(80 \mathrm{~mol} \%\) of the liquid distilled. Data From Example 6.3:- Suppose the liquid of Example 6.1 [50 mol% n-heptane (A), 50 mol% n-octane...
-
Multiple-choice questions: a. Which of the following statements is incorrect? 1. Ratios are fractions expressed in percent or times per year. 2. A ratio can be computed from any pair of numbers. 3. A...
-
What is the difference between a Relation in an RDBMS and a Class in an OODBMS? Are they interchangeable? This question is asking if a relation within a Relational Database Management System is the...
-
Consider the Everglade cash flow problem discussed in this chapter. Suppose that extra cash is kept in an interest-bearing savings account. Assume that any cash left at the end of a year earns 3...
-
Please provide necessary journal entries for the following transactions for Nathan. a) Nathan sold $2,500 worth of goods to Audrey on credit, terms 2N15, b) Audrey returned $460 worth of goods within...
-
A taxpayer has a home office of 250 square feet out of a total home size of 1,875 square feet. The office qualifies for the business use of home treatment. Expenses related to the whole home and...
-
On March 1 , 1 9 9 3 , Elwood deposited $ 4 , 2 0 0 into a bank account. The account credited interest at a nominal interest rate of 4 % per year, compounded quarterly, when the balance was under $ 5...
-
A taxpayer has $5,000 in cash donations to various charitable organizations during the year. $2,000 of these are not substantiated with a contemporaneous written acknowledgement from the donee...
-
Commercial Carpets manufactures custom mats. The details on its standard run of a 1 m x 2 m mat is as follows: Quantity Unit Selling Price Unit Variable Cost Fixed Costs 3 2 0 $ 5 6 $ 3 1 $ 1 , 9 0 0...
-
In an X-ray powder camera using incident copper radiation of wavelength 1.5418 , a diffraction line is produced at 20= 45.8. determine the miller indices for the reflecting planes if a = 3.44 .
-
Do you think organizations are using negative reinforcement more or positive reinforcement more as their means of motivating their employees? Give examples to support your position
-
Prove that the mean heat capacities C P H and C P S are inherently positive, whether T > T 0 or T < T 0 . Explain why they are well defined for T = T 0 .
-
a. State three factors that contribute to the fact that the noncatalyzed hydrolysis of an ester is a slow reaction. b. Which is faster, hydrolysis of an ester or aminolysis of the same ester?
-
Which of the following compounds are aromatic? a. b. c. Cycloheptatrienyl cation d. e. f. g. Cyclononatetraenyl anion h. CH2=CHCH=CHCH=CH2
-
Identify the following sugars: a. An aldopentose that is not D-arabinose forms D-arabinitol when it is reduced with NaBH4. b. A sugar forms the same osazone as D-galactose with phenylhydrazine, but...
-
This question is an extension of Exercise 10.22. Consider the data file \(m r o z\) on working wives and the model \(\ln (W A G E)=\beta_{1}+\beta_{2} E D U C+\beta_{3} E X P E R+e\). Use the 428...
-
Consider the data file \(m r o z\) on working wives. Use the 428 observations on married women who participate in the labor force. In this exercise, we examine the effectiveness of alternative...
-
To examine the quantity theory of money, Brumm (2005) ["Money Growth, Output Growth, and Inflation: A Reexamination of the Modern Quantity Theory's Linchpin Prediction," Southern Economic Journal,...

Study smarter with the SolutionInn App


