The coldest seawater ever recorded (in a liquid state) was in a stream under an Antarctic...
Fantastic news! We've Found the answer you've been seeking!
Question:

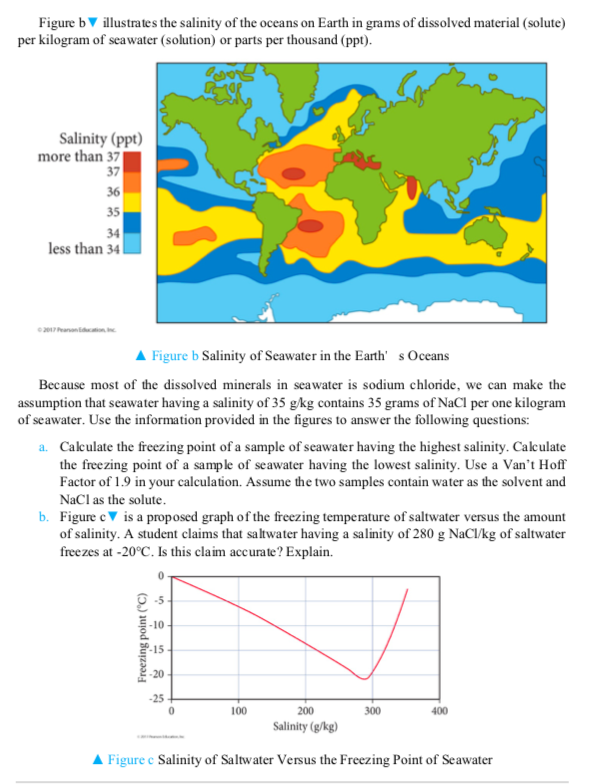

Transcribed Image Text:
The coldest seawater ever recorded (in a liquid state) was in a stream under an Antarctic glacier in 2010; it measured -2.6 °C (27.3 °F). On average, water in the world's oceans has a salinity of about 3.5% (35 g/L, or 599 mm). This means that every kilogram (roughly one liter by volume) of seawater contains approximately 35 grams (1.2 ounces) of dissolved salts (predominantly sodium (Na) and chloride (CI) ions). Figure a provides information on the amounts of various cations and anions in seawater with an average density of 1.025 g/ml. Sea Salts Seawater Sulfate- 7.7% (2.7g) Calcium- 1.2% (0.42g) Potassium- 1.1% (0.39g) Chloride 55% (19.25g) Sodium 30.6% (10.7g) Magnesium 3.7% (1.3g) -Minor constituents 0.7% (0.25g) Water 96.5% (965g) Salt 3.5% (35g) A Figure a Cations and anions in Ocean Water Figure by illustrates the salinity of the oceans on Earth in grams of dissolved material (solute) per kilogram of seawater (solution) or parts per thousand (ppt). Salinity (ppt) more than 371 37 36 35 34 less than 34 © 2017 Pearson Education, Inc. Figure b Salinity of Seawater in the Earth' s Oceans Because most of the dissolved minerals in seawater is sodium chloride, we can make the assumption that seawater having a salinity of 35 g/kg contains 35 grams of NaCl per one kilogram of seawater. Use the information provided in the figures to answer the following questions: a. Calculate the freezing point of a sample of seawater having the highest salinity. Calculate the freezing point of a sample of seawater having the lowest salinity. Use a Van't Hoff Factor of 1.9 in your calculation. Assume the two samples contain water as the solvent and NaCl as the solute. b. Figure c is a proposed graph of the freezing temperature of saltwater versus the amount of salinity. A student claims that saltwater having a salinity of 280 g NaCl/kg of saltwater freezes at -20°C. Is this claim accurate? Explain. Freezing point (°C) -10 POT 198 -15- -20 100 300 400 200 Salinity (g/kg) ▲ Figure e Salinity of Saltwater Versus the Freezing Point of Seawater c. Use the information provided in Figure b and redraw the graph in Figure c using a range of salinity values from six different oceans. The y-axis should be the freezing point of water, and the x-axis should be the salinity of seawater in units of grams of NaCl/kg of seawater. Does Figure e provide an accurate view of the freezing point of the oceans? The coldest seawater ever recorded (in a liquid state) was in a stream under an Antarctic glacier in 2010; it measured -2.6 °C (27.3 °F). On average, water in the world's oceans has a salinity of about 3.5% (35 g/L, or 599 mm). This means that every kilogram (roughly one liter by volume) of seawater contains approximately 35 grams (1.2 ounces) of dissolved salts (predominantly sodium (Na) and chloride (CI) ions). Figure a provides information on the amounts of various cations and anions in seawater with an average density of 1.025 g/ml. Sea Salts Seawater Sulfate- 7.7% (2.7g) Calcium- 1.2% (0.42g) Potassium- 1.1% (0.39g) Chloride 55% (19.25g) Sodium 30.6% (10.7g) Magnesium 3.7% (1.3g) -Minor constituents 0.7% (0.25g) Water 96.5% (965g) Salt 3.5% (35g) A Figure a Cations and anions in Ocean Water Figure by illustrates the salinity of the oceans on Earth in grams of dissolved material (solute) per kilogram of seawater (solution) or parts per thousand (ppt). Salinity (ppt) more than 371 37 36 35 34 less than 34 © 2017 Pearson Education, Inc. Figure b Salinity of Seawater in the Earth' s Oceans Because most of the dissolved minerals in seawater is sodium chloride, we can make the assumption that seawater having a salinity of 35 g/kg contains 35 grams of NaCl per one kilogram of seawater. Use the information provided in the figures to answer the following questions: a. Calculate the freezing point of a sample of seawater having the highest salinity. Calculate the freezing point of a sample of seawater having the lowest salinity. Use a Van't Hoff Factor of 1.9 in your calculation. Assume the two samples contain water as the solvent and NaCl as the solute. b. Figure c is a proposed graph of the freezing temperature of saltwater versus the amount of salinity. A student claims that saltwater having a salinity of 280 g NaCl/kg of saltwater freezes at -20°C. Is this claim accurate? Explain. Freezing point (°C) -10 POT 198 -15- -20 100 300 400 200 Salinity (g/kg) ▲ Figure e Salinity of Saltwater Versus the Freezing Point of Seawater c. Use the information provided in Figure b and redraw the graph in Figure c using a range of salinity values from six different oceans. The y-axis should be the freezing point of water, and the x-axis should be the salinity of seawater in units of grams of NaCl/kg of seawater. Does Figure e provide an accurate view of the freezing point of the oceans?
Expert Answer:
Answer rating: 100% (QA)
A Calculating the freezing point of a sample of seawater with the highest salinity 37 ppt and the lo... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemical engineering questions
-
A and B react to produce C according to the following chemical equation: A+ B C Amounts of A and B are added to an equilibrium reaction mixture of A, B, and C such that when equilibrium is again...
-
A and B disjoint, B and C not disjoint, A and C not disjoint. Draw Venn diagrams with sets A, B, and C satisfying the above requirements.
-
A and B disjoint, B and C disjoint, A and C not disjoint. Draw Venn diagrams with sets A, B, and C satisfying the above requirements.
-
Independent Challenge 1 You are the operations manager for the Chicago Arts Alliance. Each year the group revisits the number and types of activities they support to better manage their budgets. For...
-
1. Has your firm participated in any mergers or acquisitions in the past three years? What was the nature of these actions? Did they result in a consolidation of competitors? 2. Research what...
-
The following trial balance has been extracted from the ledger of Mr Yousef, a sole trader. The following additional information as at 31 May 2016 is available: (a) Rent is accrued by 210. (b) Rates...
-
Discuss the sampling strategy and technique to be used. Develop a hypothetical research scenario that would necessitate the use of the Action Research Approach and a Posttest With a Historical...
-
Below are the transactions for Ute Sewing Shop for March, the first month of operations. March 1 Issue common stock in exchange for cash of $3,000. March 3 Purchase sewing equipment by signing a note...
-
An 80 kg person stands in an elevator which is ascending towards the top of a building. A) As the elevator begins its ascent, it accelerates at the rate of 1 m/s2 . Draw the free body diagram showing...
-
Consider a system of particles uniformly distributed in space, with a constant particle number density n 0 , and characterized by a velocity distribution function f(v) such that where K 0 is a...
-
Assume the price of one share of the market index is currently $1,756.54. Earnings are currently $82.35. We expect earnings to grow at 5.59% for the next five years and at rf = 2.55% forever after...
-
A lottery claims its grand prize is \($15\) million, payable over five years at \($3\),000,000 per year. If the first payment is made immediately, what is this grand prize really worth? Use an...
-
Calculate the present value of a $1,300 discount bond with seven years to maturity if the yield to maturity is 8%.
-
What is the formula used to calculate the yield to maturity on a 20-year coupon bond with a current yield of 12% and \($1\),000 face value that sells for \($2\),500.
-
Would $200, which is to be received in exactly one year, be worth more to you today when the interest rate is 12% or when it is 17%?
-
Suppose that the cost of a movie ticket is \($12\), and a latte costs \($6\). Why would the theater management say the cost of admission is \($12\) and not two lattes? Explain why it is more...
-
The Gross Domestic Product (GDP) for the United States can be determined from totaling expenditures on goods and services produced in a year, or it can be determined by totaling the income payments...
-
TRUE-FALSE QUESTIONS 1. In terms of preliminary analytical procedures, assume that the company has introduced a new product with a low price point and significant customer demand. The auditor would...
-
Robert Ramos (age 36) is a single taxpayer, living at 8765 Bay Dr., Monterey, CA 93940. His Social Security number is 976-23-5132. Robert's earnings and income tax withholding as the manager of a...
-
During 2012, William purchases the following capital assets for use in his catering business: New passenger automobile (September 30)........................$21,500 Baking equipment (June 30)...
-
Sherry rents her vacation home for 6 months and lives in it for 6 months during the year. Her gross rental income during the year is $4,000. Total real estate taxes for the home are $950, and...
-
Define strategic human resource management, and explain using our HR Strategy Model (see Figure 3-6 in Chapter 3) how Amazons new health and safety programs fit (or dont fit) its new strategy.
-
What other health and safety actions would you recommend for them now, and why?
-
List at least five examples of how the new health and safety actions taken by Amazon reflect what you would have recommended based on what you read in this chapter.

Study smarter with the SolutionInn App


