A blast furnance burden contains 160 kg Fe203, 54 kg SiO2, 20 kg Al203, 100 kg...
Fantastic news! We've Found the answer you've been seeking!
Question:
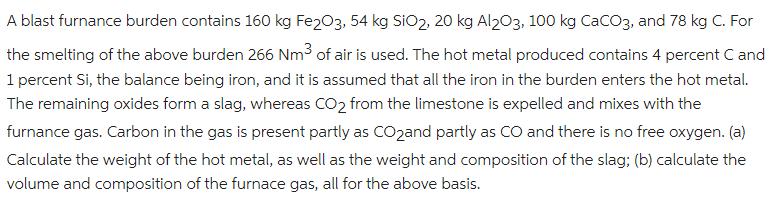
Transcribed Image Text:
A blast furnance burden contains 160 kg Fe203, 54 kg SiO2, 20 kg Al203, 100 kg CaCO3, and 78 kg C. For the smelting of the above burden 266 Nm3 of air is used. The hot metal produced contains 4 percent C and 1 percent Si, the balance being iron, and it is assumed that all the iron in the burden enters the hot metal. The remaining oxides form a slag, whereas CO2 from the limestone is expelled and mixes with the furnance gas. Carbon in the gas is present partly as COzand partly as CO and there is no free oxygen. (a) Calculate the weight of the hot metal, as well as the weight and composition of the slag; (b) calculate the volume and composition of the furnace gas, all for the above basis. A blast furnance burden contains 160 kg Fe203, 54 kg SiO2, 20 kg Al203, 100 kg CaCO3, and 78 kg C. For the smelting of the above burden 266 Nm3 of air is used. The hot metal produced contains 4 percent C and 1 percent Si, the balance being iron, and it is assumed that all the iron in the burden enters the hot metal. The remaining oxides form a slag, whereas CO2 from the limestone is expelled and mixes with the furnance gas. Carbon in the gas is present partly as COzand partly as CO and there is no free oxygen. (a) Calculate the weight of the hot metal, as well as the weight and composition of the slag; (b) calculate the volume and composition of the furnace gas, all for the above basis.
Expert Answer:

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these accounting questions
-
If it is assumed that all poker hands are equally likely, what is the probability of being dealt (a) A flush? (A hand is said to be a flush if all 5 cards are of the same suit.) (b) One pair? (This...
-
A mixture containing only Al2O3 (FM 101.96) and Fe2O3 (FM 159.69) weighs 2.019 g. When heated under a stream of H2, Al2O3 is unchanged, but Fe2O3 is converted into metallic Fe plus H2O(g). If the...
-
A cylinder/piston contains 1 kg methane gas at 100 kPa, 20C. The gas is compressed reversibly to a pressure of 800 kPa. Calculate the work required if the process is a. Adiabatic b....
-
Gabriele Enterprises has bonds on the market making annual payments, with seven years to maturity, a par value of $1,000, and selling for $974. At this price, the bonds yield 7.2 percent. What must...
-
Complete the check stub for the check you wrote in Exercise 4 if the balance brought forward is $2, 213.56 (Figure 4 - 22). Figure 4 - 22 3215 Amount To For Date 20- Balance Forward Deposits Total...
-
American Products is concerned about managing cash efficiently. On the average, inventories have an age of 90 days, and accounts receivable are collected in 60 days. Accounts payable are paid...
-
What is a compilation unit?
-
The following items were selected from among the transactions completed by Aston Martin Inc. during the current year: Apr. 15. Borrowed $225,000 from Audi Company, issuing a 30-day, 6% note for that...
-
x-4 + 3x+21 Fully simplify the NPV's. x-8x+16 x2 +12x+35 expression below. Don't forget to list your
-
The financial statements of JJ Ltd and KK Ltd for the year to 30 June 2018 are shown below: Statements of comprehensive income for the year to 30 June 2018. Statements of financial position as at 30...
-
Clark Industries has a defined benefit pension plan that specifies annual, year-end retirement benefits equal to: 15% Service years Final years salary Stanley Mills was hired by Clark at the...
-
On April 2, 2017, Montana Mining Co. pays $4,364,740 for an ore deposit containing 1,489,000 tons. The company installs machinery in the mine costing $150,600, with an estimated seven-year life and...
-
When you spread oil (nol 1.50) on water (water 1.33) and have white light incident from above, you might observe different reflected colors that depend on the thickness of the oil on the water at...
-
Mary Matthews made $950 during a biweekly pay period only social security (fully taxable) and federal income taxes attach to her pay. Matthews contributes $100 each biweekly pay to her company's...
-
Littleton Books has the following transactions during May. May 2 Purchases books on account from Readers Wholesale for $2,500, terms 2/10, n/30. May 3 Pays cash for freight costs of $120 on books...
-
Suppose that last year, the market price for a certain bond was $ 1 0 , 9 5 7 . Since then, the price has decreased by 1 0 . 9 % . If the current yield was 6 % last year, what is the current yield...
-
how do organizational development practitioners build trust between two organizations? explain
-
The baseball player A hits the ball from a height of 3.36 ft with an initial velocity of 34.8 ft/s. 0.14 seconds after the ball is hit, player B who is standing 15 ft away from home plate begins to...
-
Determine the adiabatic saturation temperature of the humid air in Prob. 14-43E. Prob. 14-43E Atmospheric air at a pressure of 1 atm and drybulb temperature of 90oF has a dew-point temperature of...
-
Consider the supersonic flow of air at upstream conditions of 70 kPa and 260 K and a Mach number of 2.4 over a two-dimensional wedge of half-angle 108. If the axis of the wedge is tilted 250 with...
-
Hydrogen (H2) is burned with 100 percent excess air that enters the combustion chamber at 80oF, 14.5 psia, and 60 percent relative humidity. Assuming complete combustion, determine (a) The air-fuel...
-
Galinha-Esquina SA grows and processes chickens. Each chicken is disassembled into five main parts. Information pertaining to production in July 2022 is as follows: Joint costs of production in July...
-
What is a joint cost?
-
Terms a Split-off point b Joint cost c Separable cost d By-product e Joint product f Product Definitions 1 Product with low sales value compared with the sales value of the main or joint product(s)....

Study smarter with the SolutionInn App


