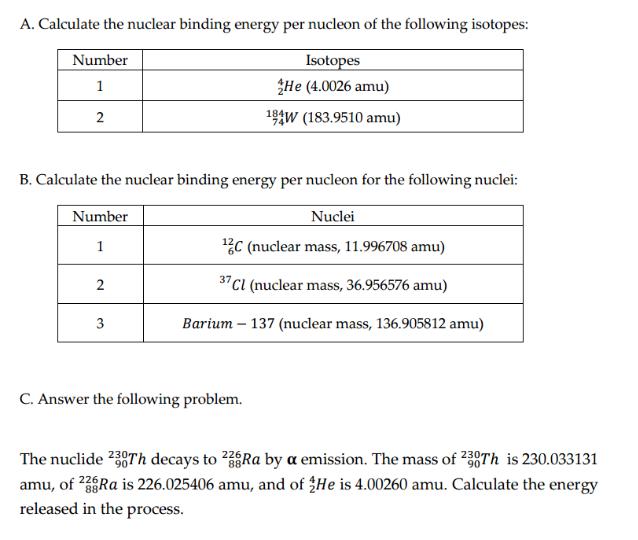
A. Calculate the nuclear binding energy per nucleon of the following isotopes: Isotopes He (4.0026 amu)...
Fantastic news! We've Found the answer you've been seeking!
Question:
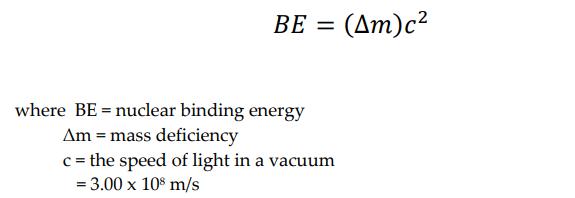
Transcribed Image Text:
A. Calculate the nuclear binding energy per nucleon of the following isotopes: Isotopes He (4.0026 amu) 194W (183.9510 amu) Number 1 2 B. Calculate the nuclear binding energy per nucleon for the following nuclei: Number Nuclei 1 ¹2C (nuclear mass, 11.996708 amu) 37 Cl (nuclear mass, 36.956576 amu) Barium - 137 (nuclear mass, 136.905812 amu) 2 3 C. Answer the following problem. 226 The nuclide 230Th decays to 238Ra by a emission. The mass of 230Th is 230.033131 amu, of 23Ra is 226.025406 amu, and of He is 4.00260 amu. Calculate the energy released in the process. BE = (Δm)c2 where BE = nuclear binding energy Am = mass deficiency c = the speed of light in a vacuum = 3.00 x 108 m/s A. Calculate the nuclear binding energy per nucleon of the following isotopes: Isotopes He (4.0026 amu) 194W (183.9510 amu) Number 1 2 B. Calculate the nuclear binding energy per nucleon for the following nuclei: Number Nuclei 1 ¹2C (nuclear mass, 11.996708 amu) 37 Cl (nuclear mass, 36.956576 amu) Barium - 137 (nuclear mass, 136.905812 amu) 2 3 C. Answer the following problem. 226 The nuclide 230Th decays to 238Ra by a emission. The mass of 230Th is 230.033131 amu, of 23Ra is 226.025406 amu, and of He is 4.00260 amu. Calculate the energy released in the process. BE = (Δm)c2 where BE = nuclear binding energy Am = mass deficiency c = the speed of light in a vacuum = 3.00 x 108 m/s
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these physics questions
-
The isotope has the largest binding energy per nucleon of any isotope. Calculate this value from the atomic mass of nickel-62 (61.928345 amu) and compare it with the value given for iron-56 in Table...
-
Calculate the nuclear binding energy (in J) and the binding energy per nucleon of the following isotopes: (a) 73Li (7.01600 amu) and (b) 3517Cl(34.95952 amu).
-
Sound waves with frequency 3000 Hz and speed 343 m/s diffract through the rectangular opening of a speaker cabinet and into a large auditorium of length d = 100 m. The opening, which has a horizontal...
-
Pam recently was sickened by eating spoiled peanut butter. She successfully sued the manufacturer for her medical bills ($3,700), her emotional distress ($6,000 she now fears peanut butter), and...
-
Consider the following sample data: a. Find the sample regression equation, = b0 + b1x1 + b2x2. b. Construct the 95% confidence interval for E(y) if x1 equals 50 and x2 equals 20. c. Construct the...
-
You are the global sales manager for a consumer food products company that is getting ready to roll out a new product. As part of the introduction your global sales force is being asked to do a...
-
As HR manager at Krameria, Inc., you are trying to determine whether there is a useful linear relationship between x, the years of education of a company employee, and y, the employees job...
-
HR manager is looking into employee absence. 10% of all plant employees work in the finishing department; 20% of all plant employees are absent excessively; and 7% of all plant employees work in the...
-
Use PSpice to find V1, V2, and V3 in the network of Fig. 10.128. 8 V. j1012 V, jlOS2 4/0 A
-
Travel websites such as Expedia and Priceline tend to offer reservation services for multiple travel modes. Based only on your observation of this trend, provide an explanation that is most plausible...
-
True or False: When dealing with multiple alternatives having unequal lives, the planning horizon equals the least common multiple of lives.
-
True or False: The MARR should be at least as great as the firm's weighted cost of capital and should reflect the opportunity cost for money.
-
True or False: The cost of debt capital needs to reflect the tax deductibility of costs of debt.
-
True or False: The Eastman hurdle rate calculator can be used for any company and under any conditions that are specifically targeted by the calculator.
-
True or False: Among the various sources of capital, U.S. large cap equities have a lower cost of capital than do U.S. small cap equities.
-
Conduct an internet search on age discrimination. Choose three sites that each deal with a different aspect of age discrimination (for example: discrimination in high tech industries, preventing...
-
How has the too-big-to-fail policy been limited in the FDICIA legislation? How might limiting the too-big-to-fail policy help reduce the risk of a future banking crisis?
-
A 0.040 M solution of a monoprotic acid is 14 percent ionized. Calculate the ionization constant of the acid.
-
Astatine, the last member of Group 7A, can be prepared by bombarding bismuth-209 with particles. (a) Write an equation for the reaction. (b) Represent the equation in the abbreviated form, as...
-
A concentration of 8.00 102 ppm by volume of CO is considered lethal to humans. Calculate the minimum mass of CO in grams that would become a lethal concentration in a closed room 17.6 m long, 8.80...
-
Which of the following is not a reason that the prevention and detection of fraud resulting from management override and collusion presents a significant challenge for the antifraud community? 1....
-
Which of the following statements best describes corporate governance with respect to fraud? 1. Auditors are primarily responsible for the detection of fraud, the Board of Directors for the...
-
Which of the following is not an inherent part of Statement on Auditing Standards, No. 99/113? 1. Greater scrutiny of the chief executive and chief financial officers personal financial condition 2....

Study smarter with the SolutionInn App


