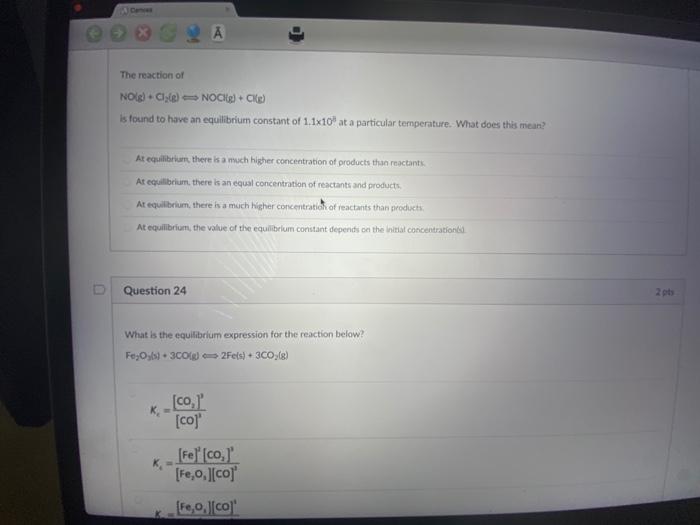
A Conver The reaction of NO(g) + Cl(g) NOCK(g) + CKg) is found to have an...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
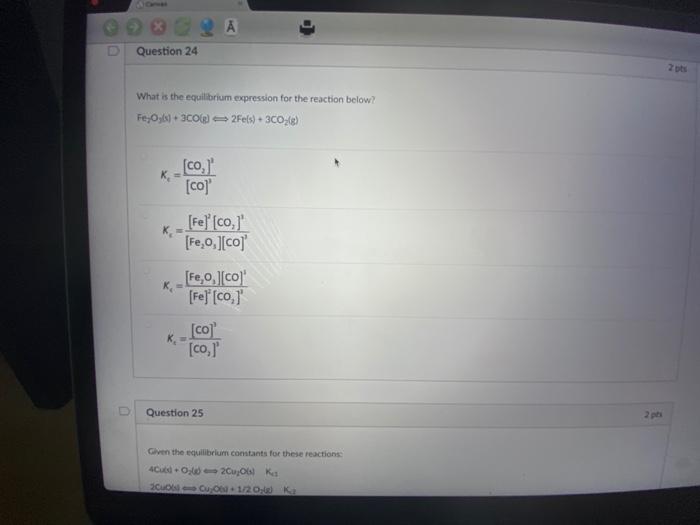
A Conver The reaction of NO(g) + Cl(g) NOCK(g) + CKg) is found to have an equilibrium constant of 1.1x10 at a particular temperature. What does this mean? 2 A At equilibrium, there is a much higher concentration of products than reactants At equilibrium, there is an equal concentration of reactants and products. At equilibrium, there is a much higher concentration of reactants than products. At equilibrium, the value of the equilibrium constant depends on the initial concentrationé D Question 24 What is the equilibrium expression for the reaction below? Fe₂Oys) + 3C0(g) 2Fe(s) + 3CO; (g) K₂ K [co, [co]' [Fe] [co, [Fe,o,][co] [Fe,0,][co] 2 pts Question 24 What is the equilibrium expression for the reaction below? Fe,Oys) + 3C0(g) 2Fe(s) + 3CO₂(g) _[co,]' [co]' [Fe] [co,]' [Fe₂O,][co] K [Fe,o,][co]' [Fe] [co,]' [co] [co,]' Question 25 Given the equilibrium constants for these reactions: 2Cu₂06) Kes 4Cubd-O 20u06) Cu 06-1/20g) Ka 2 pts 2 pts A Conver The reaction of NO(g) + Cl(g) NOCK(g) + CKg) is found to have an equilibrium constant of 1.1x10 at a particular temperature. What does this mean? 2 A At equilibrium, there is a much higher concentration of products than reactants At equilibrium, there is an equal concentration of reactants and products. At equilibrium, there is a much higher concentration of reactants than products. At equilibrium, the value of the equilibrium constant depends on the initial concentrationé D Question 24 What is the equilibrium expression for the reaction below? Fe₂Oys) + 3C0(g) 2Fe(s) + 3CO; (g) K₂ K [co, [co]' [Fe] [co, [Fe,o,][co] [Fe,0,][co] 2 pts Question 24 What is the equilibrium expression for the reaction below? Fe,Oys) + 3C0(g) 2Fe(s) + 3CO₂(g) _[co,]' [co]' [Fe] [co,]' [Fe₂O,][co] K [Fe,o,][co]' [Fe] [co,]' [co] [co,]' Question 25 Given the equilibrium constants for these reactions: 2Cu₂06) Kes 4Cubd-O 20u06) Cu 06-1/20g) Ka 2 pts 2 pts
Expert Answer:
Answer rating: 100% (QA)
Question23 Solution The given chemical reaction is as follows NO g Cl 2 g NOCl g Cl g The value of t... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A diatomic molecule is found to have an activation energy of 1.4eV. When the molecule is disassociated, 1.6eV of energy is released. Draw a potential energy curve for this molecule.
-
A hypothetical element X is found to have an atomic mass of 37.45 amu. Element X has only two isotopes, X-37 and X-38. The X-37 isotope has a fractional abundance of 0.7721 and an isotopic mass of...
-
A diatomic molecule is found to have an activation energy of 1.3 eV. When the molecule is disassociated, 1.6 eV of energy is released. Draw a potential energy curve for this molecule.
-
Figure 2 shows a 250 kg beam BC at the time instant when a 100 N horizontal force is applied to its end B for 10 s. a. Determine the tension in rods AB and CD at an arbitrary instant during the time...
-
How is the unrealized gross profit on intra-entity sales calculated? What effect does an unrealized gross profit have on the recording of an investment if the equity method is applied?
-
For the past five years, Garner Company has had a policy of producing to meet customer demand. As a result, finished goods inventory is minimal, and for the most part, units produced equal units...
-
True or False: If \(P W>0\), then \(I R R>M A R R\).
-
Target operating income, value-added costs, service company. Calvert Associates prepares architectural drawings to conform to local structural-safety codes. Its income statement for 2012 is as...
-
A 25-year-old Latin, G1P1, cisfemale presents to the office with a 6-month history of amenorrhea; reports having regular menstrual cycles every month up until about 1 year ago when her menstrual...
-
Ellery Products manufactures various components for the fashion industry. Ellery buys fabric from two vendors: Ewers Textiles and Bramford Materials. Ellery chooses the vendor based on price. Once...
-
Write a detailed analyzed paper about Mount Sinai Medical Hospital
-
Comment on the advantages of the DB approach to project delivery that the Design-Build Institute of America (DBIA) provides on its website (www.dbia.org). Does the DB approach have any potential...
-
What is meant by the following terms? a. Yellow-dog contract b. Agency shop c. Subcontractor clause
-
Suppose in the preceding problem that the bonds are financed by a bank that discounts the bond issue to $40 million. What is the new effective interest rate?
-
What is a secondary boycott? Name two types of secondary boycotts. Does the legislation forbidding secondary boycotts apply to construction unions? Explain.
-
You have just bought a used track-type tractor to add to your production fleet. The initial capitalized value of the tractor is $110,000. The estimated service life remaining on the tractor is 10,000...
-
4 Following the acquisition of ZZA company during Year 20X8, ABC Corporation released its Year 20X8 financial statements. The Year 20X8 financial statements and other data are reproduced below. ABC...
-
Conduct a VRIO analysis by ranking Husson University (in Maine) business school in terms of the following six dimensions relative to the top three rival schools. If you were the dean with a limited...
-
What is the total charge of all the electrons in a 25-kg bar of aluminum? (Aluminum has 13 electrons per atom and an atomic mass of 27 u.)
-
Which of the following statements is true? (a) A larger-diameter lens can better resolve two distant points. (b) Red light can better resolve two distant points than blue light can. (c) It is easier...
-
Estimate the peak wavelength of light emitted from the pupil of the human eye (which approximates a blackbody) assuming normal body temperature.
-
On November 15, 20X3, Chow Inc., a U.S. company, ordered merchandise FOB shipping point from a German company for 200,000. The merchandise was shipped and invoiced on December 10, 20X3. Chow paid the...
-
On April 8, 20X3, Trul Corporation purchased merchandise from an unaffiliated foreign company for 10,000 units of the foreign companys local currency. Trul paid the bill in full on March 1, 20X4,...
-
On September 1, 20X1, Cott Corporation received an order for equipment from a foreign customer for 300,000 LCUs when the U.S. dollar equivalent was $96,000. Cott shipped the equipment on October 15,...

Study smarter with the SolutionInn App


