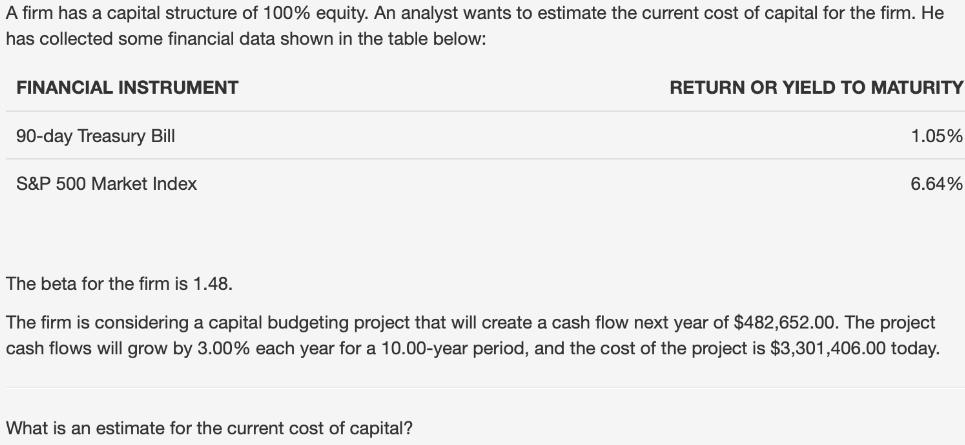
A firm has a capital structure of 100% equity. An analyst wants to estimate the current...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A firm has a capital structure of 100% equity. An analyst wants to estimate the current cost of capital for the firm. He has collected some financial data shown in the table below: FINANCIAL INSTRUMENT 90-day Treasury Bill S&P 500 Market Index RETURN OR YIELD TO MATURITY What is an estimate for the current cost of capital? 1.05% 6.64% The beta for the firm is 1.48. The firm is considering a capital budgeting project that will create a cash flow next year of $482,652.00. The project cash flows will grow by 3.00% each year for a 10.00-year period, and the cost of the project is $3,301,406.00 today. A firm has a capital structure of 100% equity. An analyst wants to estimate the current cost of capital for the firm. He has collected some financial data shown in the table below: FINANCIAL INSTRUMENT 90-day Treasury Bill S&P 500 Market Index RETURN OR YIELD TO MATURITY What is an estimate for the current cost of capital? 1.05% 6.64% The beta for the firm is 1.48. The firm is considering a capital budgeting project that will create a cash flow next year of $482,652.00. The project cash flows will grow by 3.00% each year for a 10.00-year period, and the cost of the project is $3,301,406.00 today.
Expert Answer:
Answer rating: 100% (QA)
Capital cost can be calculated using the Capital Asset Pricing Model CAPM The CAPM equation is Cost of Equity RiskFree Rate plus Beta multiplied by the Market Risk Premium Utilize code with care Find ... View the full answer

Related Book For 

Financial Reporting Financial Statement Analysis and Valuation a strategic perspective
ISBN: 978-1337614689
9th edition
Authors: James M. Wahlen, Stephen P. Baginski, Mark Bradshaw
Posted Date:
Students also viewed these finance questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
An analyst wants to evaluate portfolio X, consisting entirely of U.S. common stocks, using both the Treynor and Sharpe measures of portfolio performance. The following table provides the average...
-
A group of investors is intent on purchasing a publicly traded company and wants to estimate the highest price they can reasonably justify paying. The target companys equity beta is 1.20 and its...
-
Give the three ways to eliminate the balance in the overhead account at the end of the year.
-
A cylindrical resistor of radius 5.0 mm and length 2.0 cm is made of material that has a resistivity of 3.5 x 10-5 m. What are? (a) The magnitude of the current density and (b) The potential...
-
Which of these processes are nonspontaneous? Are the nonspontaneous processes impossible? a. A bike going up a hill b. A meteor falling to Earth c. Obtaining hydrogen gas from liquid water d. A ball...
-
Derive the expression for the stress induced in a shaft with an unbalanced concentrated mass located midway between two bearings.
-
Following are audit procedures commonly performed in the inventory and warehousing cycle for a manufacturing company: 1. Read the client's physical inventory instructions and observe whether they are...
-
Speeding up and slowing down A 1400kg car, starting from rest, accelerates to a speed of 30mph in 10 seconds. a) What is the force on the car (assumed constant) over this period of time? b) Where...
-
You work for Euroc, a limited liability company, which seeks growth through acquisitions. You are a member of a team that is investigating the possible purchase of Choggerell, a limited liability...
-
the endowment effect, loss aversion, range effects, reference dependence, reward prediction error, failure to consider opportunity costs, failure to consider sunk costs. Which of these phenomenon...
-
Create multiple estimates of the intrinsic value of equity for Ford Tesla, Mazda and Volvo, including the use of a dividend discount, model and method of comparable. Compare your estimated values to...
-
Kelly James started a software development business as a sole proprietorship on 3 /1/XX. During the month of March the company had the following transactions: 1/1The owner invested $80,000 cash into...
-
SMCC Retail Corp had the following info: Beg inventory ~ cost = $400,000, @ retail =$790,000 Net Purchases @ cost = $1,475,000, @ retail = $2,350,000 In addition, Mark-ups were $100,000, mark-up...
-
On the first day of Year Two, the Richmond Corporation holds accounts receivable of$400,000 and an allowance for doubtful accounts of $23,000 for a net realizable value of$377,000. During the year,...
-
MedTech Ltd (MedTech) imports a number of pharmaceutical products. In order to hedge its foreign currency transactions, MedTech entered into a number of forward rate agreements this year. Prior to...
-
On August 29th, 2020, you buy a share of Emerge Energy Services Stock (EMES) after getting a hot tip. The purchase price was $144.17. After what was a bit of a rough year, you sell the stock on...
-
"Standard-cost procedures are particularly applicable to process-costing situations." Do you agree? Why?
-
Visa Inc., a credit card company, reported income tax expense of $1,648 million for Year 1, comprising $1,346 million of current taxes and $302 million of deferred taxes. The balance sheet showed...
-
Suppose the following hypothetical data represent total assets, book value, and market value of common shareholders equity (dollar amounts in millions) for Abbott Labs, IBM, and Target Stores. Abbott...
-
Walmart Stores, Inc. (Walmart) is the largest retailing firm in the world. Building on a base of discount stores, Walmart has expanded into warehouse clubs and Supercenters, which sell traditional...
-
A piece of red hot iron is suddenly plunged into \(10 \mathrm{~kg}\) of water at \(20^{\circ} \mathrm{C}\). The temperature of water rises to \(55^{\circ} \mathrm{C}\). Calculate the change in...
-
A reverse Carnot engine is used for the heating of a building. The building receives \(209 \mathrm{~kJ} / \mathrm{h}\) of heat at \(20^{\circ} \mathrm{C}\) from a heat pump. The outside air...
-
A hot iron having specific heat \(0.5 \mathrm{~kJ} / \mathrm{kg} \mathrm{K}\), mass \(30 \mathrm{~kg}\) and temperature \(500^{\circ} \mathrm{C}\) is dropped into \(200 \mathrm{~kg}\) of oil of...

Study smarter with the SolutionInn App


