A food storage locker requires a refrigeration system of 50 kW capacity at an evaporator temperature...
Fantastic news! We've Found the answer you've been seeking!
Question:
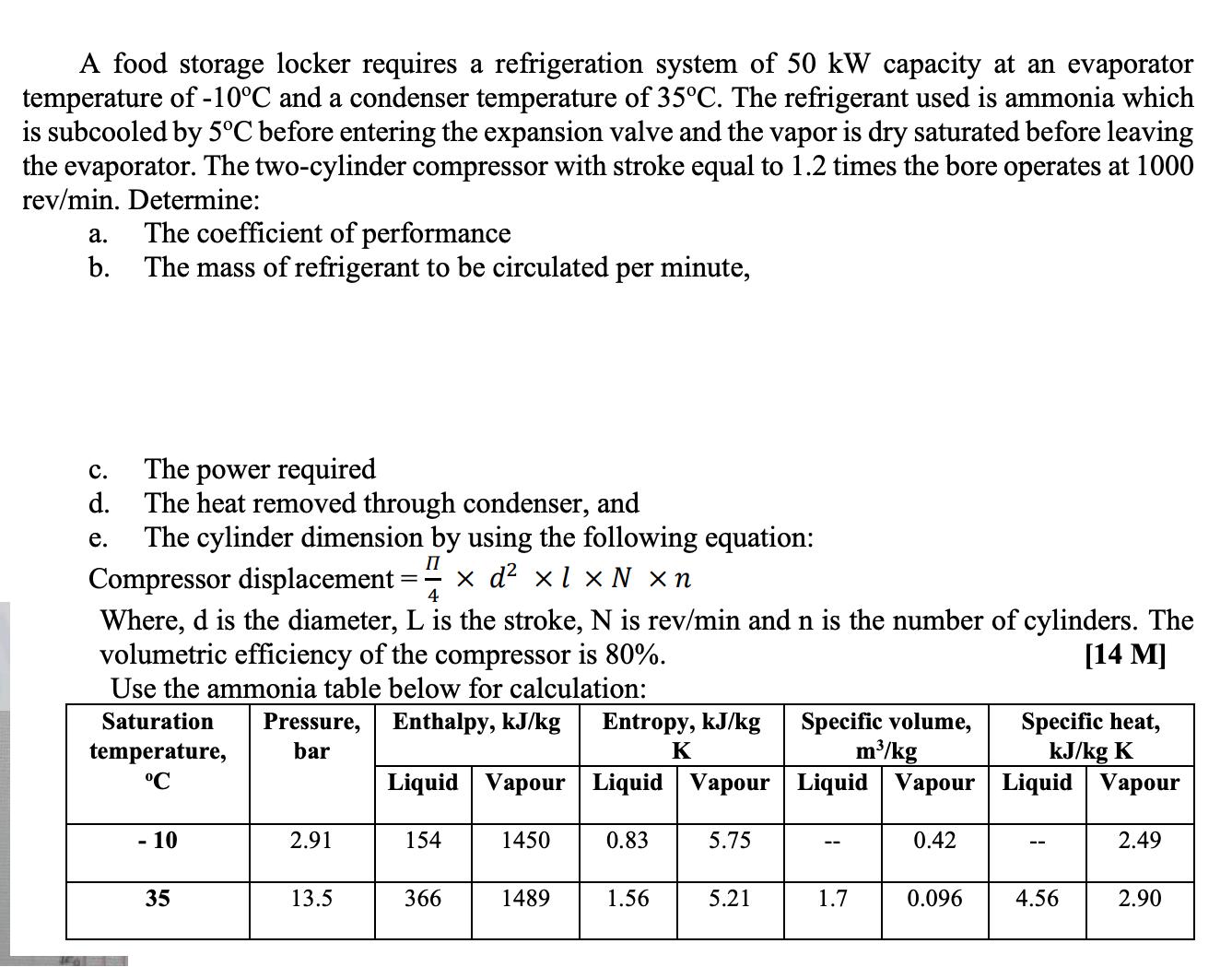
Transcribed Image Text:
A food storage locker requires a refrigeration system of 50 kW capacity at an evaporator temperature of -10°C and a condenser temperature of 35°C. The refrigerant used is ammonia which is subcooled by 5°C before entering the expansion valve and the vapor is dry saturated before leaving the evaporator. The two-cylinder compressor with stroke equal to 1.2 times the bore operates at 1000 rev/min. Determine: #al a. b. C. d. e. The coefficient of performance The mass of refrigerant to be circulated per minute, The power required The heat removed through condenser, and The cylinder dimension by using the following equation: II Compressor displacement: x d² xl x N xn 4 Where, d is the diameter, L is the stroke, N is rev/min and n is the number of cylinders. The volumetric efficiency of the compressor is 80%. [14 M] Use the ammonia table below for calculation: Enthalpy, kJ/kg Saturation temperature, °℃ - 10 35 Pressure, bar 2.91 13.5 Liquid Vapour 154 366 1450 1489 Entropy, kJ/kg K Liquid Vapour 5.75 0.83 1.56 5.21 Specific volume, m³/kg Liquid Vapour 1.7 0.42 0.096 Specific heat, kJ/kg K Liquid Vapour -- 4.56 2.49 2.90 A food storage locker requires a refrigeration system of 50 kW capacity at an evaporator temperature of -10°C and a condenser temperature of 35°C. The refrigerant used is ammonia which is subcooled by 5°C before entering the expansion valve and the vapor is dry saturated before leaving the evaporator. The two-cylinder compressor with stroke equal to 1.2 times the bore operates at 1000 rev/min. Determine: #al a. b. C. d. e. The coefficient of performance The mass of refrigerant to be circulated per minute, The power required The heat removed through condenser, and The cylinder dimension by using the following equation: II Compressor displacement: x d² xl x N xn 4 Where, d is the diameter, L is the stroke, N is rev/min and n is the number of cylinders. The volumetric efficiency of the compressor is 80%. [14 M] Use the ammonia table below for calculation: Enthalpy, kJ/kg Saturation temperature, °℃ - 10 35 Pressure, bar 2.91 13.5 Liquid Vapour 154 366 1450 1489 Entropy, kJ/kg K Liquid Vapour 5.75 0.83 1.56 5.21 Specific volume, m³/kg Liquid Vapour 1.7 0.42 0.096 Specific heat, kJ/kg K Liquid Vapour -- 4.56 2.49 2.90
Expert Answer:
Answer rating: 100% (QA)
a Coefficient of performance COP Heat removed from evaporator Work by compressor Qeva W Heat ... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these accounting questions
-
What are the sociological implications of a highly bureaucratized system on the morale and motivation of public sector employees?
-
Msucat Autos is offering great deals on the new BMW, which run for OMR 18,000 fully loaded. You pay OMR 1,000 down payment and then OMR 400 a month for the next 36 months, Al-Seeb Motors next door...
-
It is possible to construct a nonparametric tolerance interval that is based on the extreme values in a random sample of size n from any continuous population. If p is the minimum proportion of the...
-
Residents of Hawaii are warned of the approach of a tsunami by sirens mounted on the tops of towers. Suppose a siren produces a sound that has an intensity level of 120 dB at a distance of 2.0 m....
-
Why is it important to distinguish between the performance of a manager and the performance of the organization subunit for which the manager is responsible? Give examples.
-
Water flows steadily through the horizontal piping system shown in Fig. P5.3. The velocity is uniform at section (1), the mass flowrate is 10 slugs/s at section (2), and the velocity is nonuniform at...
-
Here is the condensed 2008 balance sheet for Skye Computer Company (in thousands of dollars): 2008 Current assets $ 2,000 Net fixed assets 3,000 Total assets $ 5,000 Current liabilities $ 900...
-
Read the three factual scenarios and discuss whether the actions were done under color of state law. Please give reasons in support of your conclusion. Two New York City police officers removed...
-
1. Prepare the journal entries for transactions. (If no entry is required for a particular transaction/event, select "No journal entry required" in the first account field.) 2. Prepare an income...
-
Construct several (at least 3) Voronoi polyhedra on a representative part of the structure (e.g. not along the edge). Calculate the average number of nearest neighbors for the structure based on your...
-
Compare and contrast the strengths and weaknesses of two (2) federal mitigation programs. Be sure to explain how each functions to mitigate hazards in the United States.
-
At the end of the year 2022, Statistics South Africa estimated the total amount spent on consumption, government purchases, investment, and net export of goods and services to arrive at the country's...
-
According to the Google, Inc. Privacy Policy Litigation, on what date did Google announce their new universal privacy policy?
-
What would be considered "accumulated taxable income" for the year?
-
Explain how the reasons for the downward sloping of a demand curve for a normal good can be applied to the demand for healthcare goods and services.
-
Determine the magnitude of the force P that would cause block A to accelerate to the right at 4m/s^2. Neglect friction and the mass of the pulley. I found answers from the website but I wonder why...
-
The area of a rectangle is 30 cm 2 and its perimeter is 26 cm. Find the length and width of the rectangle.
-
Reconsider Prob. 9-55. Using EES (or other) software, study the effect of varying the compression ratio from 14 to 24. Plot the net work output, mean effective pressure, and thermal efficiency as a...
-
A 1200-W electric resistance heating element whose diameter is 0.5 cm is immersed in 40 kg of water initially at 20C. Assuming the water container is well-insulated, determine how long it will take...
-
Repeat Prob. 16-86 for a relative humidity of 25 percent. Prob. 16-86 Consider a glass of water in a room at 25C and 100 kPa. If the relative humidity in the room is 25 percent and the water and the...
-
Isothermal compression efficiency can be achieved by running the compressor: (a) At a very high speed (b) At a very slow speed (c) At an average speed (d) At zero speed
-
Derive the expression of work done by the compressor in isothermal compression, adiabatic compression, and polytropic compression.
-
Maximum work is done in compressing air when the compression is: (a) Isothermal compression (b) Adiabatic compression (c) Polytropic compression (d) None of these

Study smarter with the SolutionInn App


