Question: A four-cylinder, four-stroke gasoline engine is modeled using an ideal air-standard Otto cycle consisting of the following four internally reversible processes. Process 1-2: Adiabatic
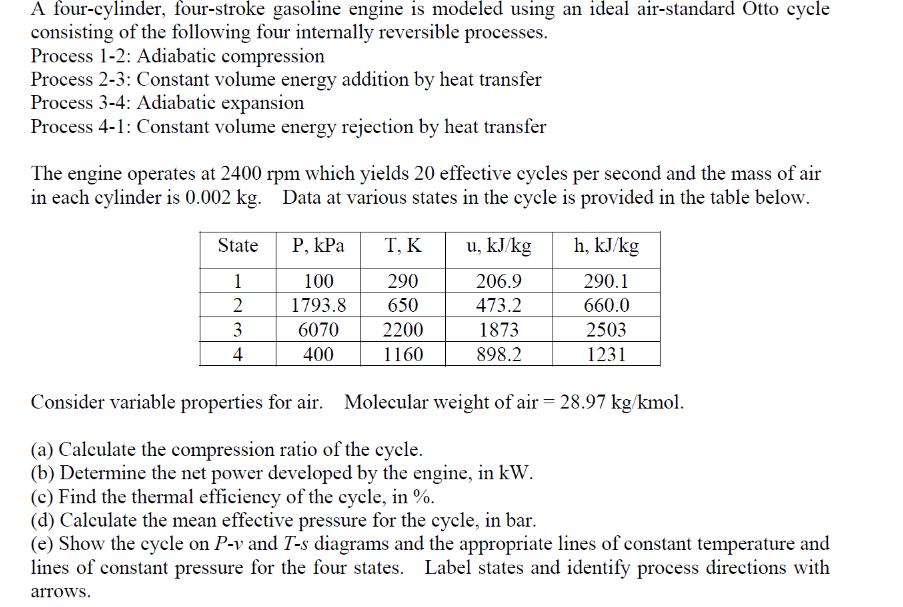
A four-cylinder, four-stroke gasoline engine is modeled using an ideal air-standard Otto cycle consisting of the following four internally reversible processes. Process 1-2: Adiabatic compression Process 2-3: Constant volume energy addition by heat transfer Process 3-4: Adiabatic expansion Process 4-1: Constant volume energy rejection by heat transfer The engine operates at 2400 rpm which yields 20 effective cycles per second and the mass of air in each cylinder is 0.002 kg. Data at various states in the cycle is provided in the table below. State 1 2 3 4 P, kPa T, K 290 100 1793.8 650 6070 2200 400 1160 u, kJ/kg 206.9 473.2 1873 898.2 h, kJ/kg 290.1 660.0 2503 1231 Consider variable properties for air. Molecular weight of air = 28.97 kg/kmol. (a) Calculate the compression ratio of the cycle. (b) Determine the net power developed by the engine, in kW. (c) Find the thermal efficiency of the cycle, in %. (d) Calculate the mean effective pressure for the cycle, in bar. (e) Show the cycle on P-v and T-s diagrams and the appropriate lines of constant temperature and lines of constant pressure for the four states. Label states and identify process directions with arrows.
Step by Step Solution
3.38 Rating (148 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


