A frictionless and leak-proof piston with a de (in kg) and negligible thickness, is in equilibrium...
Fantastic news! We've Found the answer you've been seeking!
Question:

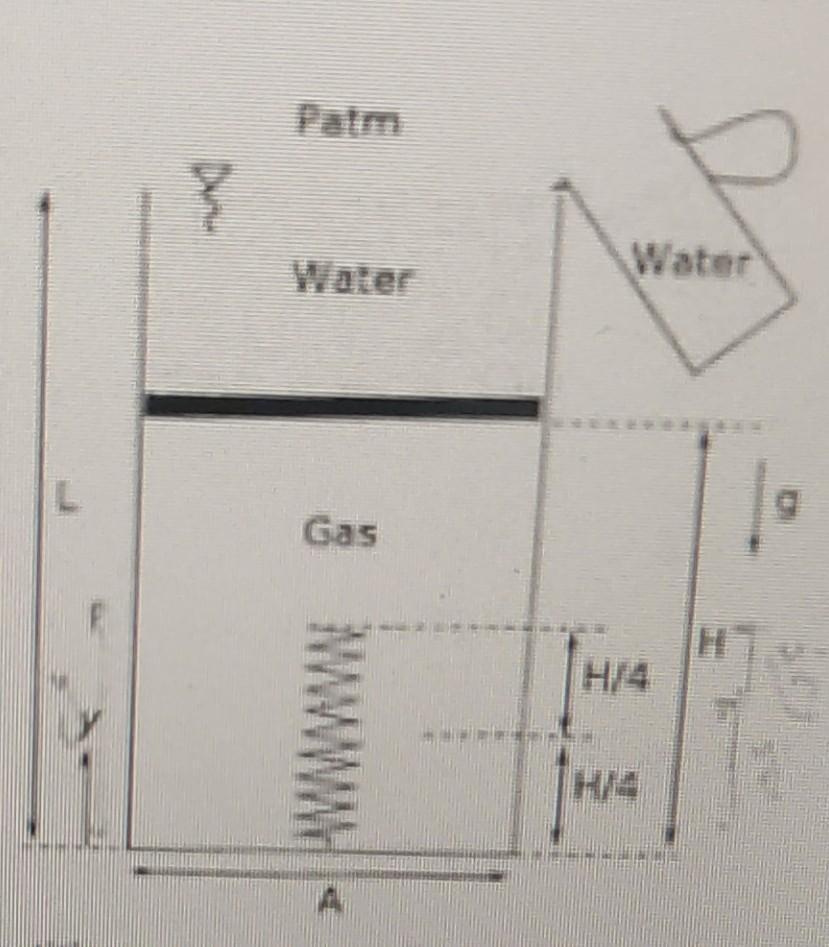
Transcribed Image Text:
A frictionless and leak-proof piston with a de (in kg) and negligible thickness, is in equilibrium as shown in the figure with gas below it and water above it till the top. The cross-sectional area of the cylinder is A (in m) and the total length is L (in m). A linear spring with a stiffness K (in N/m) is placed inside the cylinder, which is initially uncompressed, with a length of H/2 (H in meters). Water is slowly poured such that no water spills from the top of the cylinder and the piston moves down slowly to accommodate the additional water that is being added. The process of adding water is stopped when the spring is compressed by a length of H/4 Neglect the volume of the spring and take the atmospheric pressure as Patm (in Pa). Using the given variables, obtain relations for the following quantity. (a) Initial and final pressures of the gas. wh (b) Expressions for intermediate pressures of the gas bottom, for the ranges H/ 2 L D4 } Patm Water Gas H/4 H/4 A frictionless and leak-proof piston with a de (in kg) and negligible thickness, is in equilibrium as shown in the figure with gas below it and water above it till the top. The cross-sectional area of the cylinder is A (in m) and the total length is L (in m). A linear spring with a stiffness K (in N/m) is placed inside the cylinder, which is initially uncompressed, with a length of H/2 (H in meters). Water is slowly poured such that no water spills from the top of the cylinder and the piston moves down slowly to accommodate the additional water that is being added. The process of adding water is stopped when the spring is compressed by a length of H/4 Neglect the volume of the spring and take the atmospheric pressure as Patm (in Pa). Using the given variables, obtain relations for the following quantity. (a) Initial and final pressures of the gas. wh (b) Expressions for intermediate pressures of the gas bottom, for the ranges H/ 2 L D4 } Patm Water Gas H/4 H/4
Expert Answer:

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these physics questions
-
Low Country Goods has four employees and pays them on an hourly basis. During the week beginning June 2 4 and ending June 3 0 , these employees worked the hours shown below. Information about hourly...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
A piston/cylinder with cross sectional area of 0.01 m2 has a piston mass of 100 kg resting on the stops, as shown in Fig. P2.45. with an outside atmospheric pressure of 100 kPa, what should the water...
-
Last year, Lena biked 330 miles. This year, she biked k miles. Using k, write an expression for the total number of miles she biked.
-
Given the stress state in Problem 1.11, find: the magnitude and direction of the maximum shear stress and illustrate with a sketch.
-
A 2-mm thick piece of sheet steel is cut and bent into the machine component shown. Knowing that the density of steel is 7850 kg/m3, determine the mass moment of inertia of the component with respect...
-
A single cylinder, four stroke cycle oil engine is fitted with a rope brake. The diameter of the brake wheel is \(600 \mathrm{~mm}\) and rope diameter is \(26 \mathrm{~mm}\). The dead load on the...
-
Kenneth and Patricia Golding spent a career as a husband-and-wife real estate investment partnership in Washington, DC. When they finally retired to a 25-acre farm in northern Virginias Fairfax...
-
Explore the principles of statistical process control (SPC) in manufacturing systems, discussing the use of control charts, process capability analysis, and process performance indices to monitor,...
-
You work in the human resources department of your company helping new employees fill out the necessary paperwork to get their first paycheck. There are a number of decisions that employees must make...
-
Assume that you are a hotel manager. The food buyer and chef are blaming each other for the low quality of steak dinners. The chef claims that the buyer is purchasing inferior meat. The buyer...
-
We witnessed a historic event in the commodity market in April 2020. The oil futures price plunged into negative. There are many reasons behind this puzzling phenomenon. Does our formula For=Soe(r-)T...
-
In preparing a Statement of Cash Flows, you encountered the following transaction of February 1, 2013: acquired a small office building in exchange for 5,000 of our common shares; market value $15...
-
Using the strain-based Morrow fatigue equation (Combination of Coffin-Manson relation and Basquin relation) for combined LCF-HCF fatigue model, find the max allowable strain amplitude for completely...
-
2. Consider the following Industry Demand and Total Cost Curves for an industry characterized by monopoly: Total Cost 650,000 + 8Q Demand: P = 1,084,000 - 152Q a) What are the marginal cost, average...
-
These two compression utilities compress files and are similar in their functions and operations. The main difference is that compression than , but the latter is much more widely used offers...
-
Consider a uniform gravitational field (a fair approximation near the surface of a planet). Find where Express your answer in terms of m, g, yo, and yf. View Available Hint(s) U(yf) - U (yo) =...
-
A regular deposit of $100 is made at the beginning of each year for 20 years. Simple interest is calculated at i% per year for the 20 years. At the end of the 20-year period, the total interest in...
-
Air flowing at 8 psia, 480 R, and Ma1 5 2.0 is forced to undergo a compression turn of 158. Determine the Mach number, pressure, and temperature of air after the compression.
-
Consider a simple ideal Brayton cycle operating between the temperature limits of 300 and 1500 K. Using constant specific heats at room temperature, determine the pressure ratio for which the...
-
Atmospheric contaminants are often measured in parts per million (by volume). What would the partial pressure of refrigerant-134a be in atmospheric air at 100 kPa and 20oC to form a 100-ppm...
-
How does risk affect the financial management of sport organizations?
-
Describe the process of determining a nominal interest rate.
-
Of MLB, the NBA, and the NHL, which league has the most risk, and which has the least? Why?

Study smarter with the SolutionInn App


