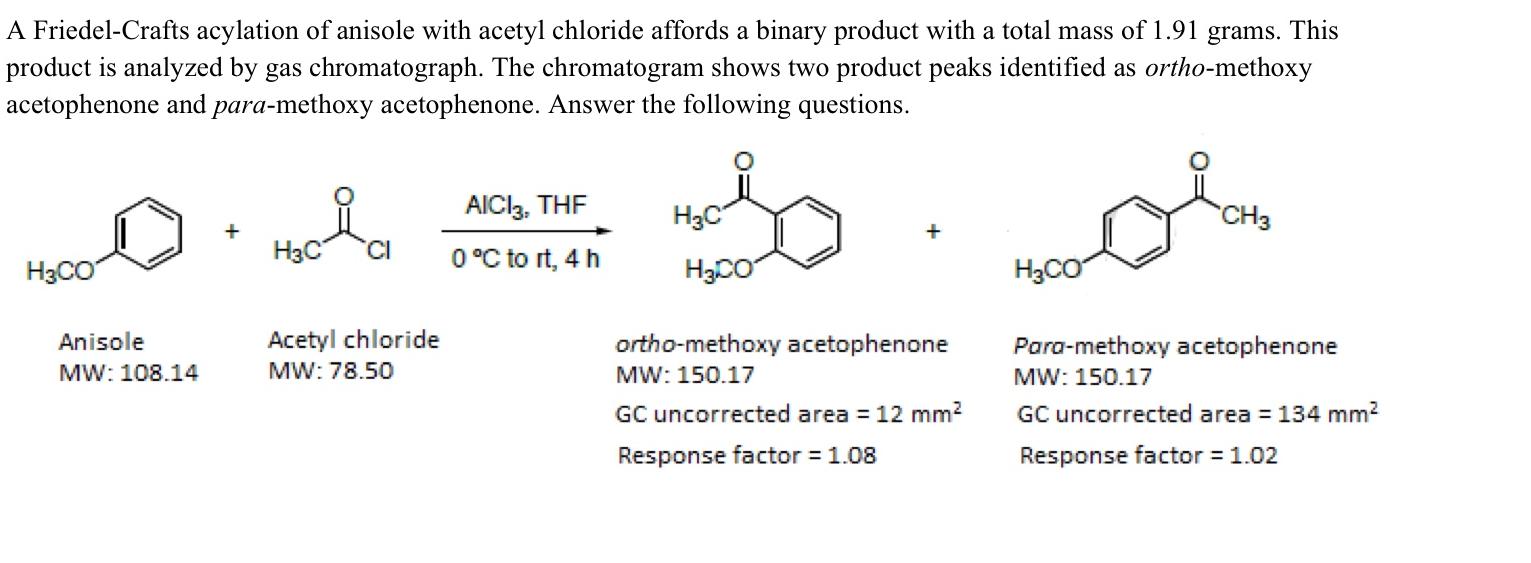
A Friedel-Crafts acylation of anisole with acetyl chloride affords a binary product with a total mass...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
A Friedel-Crafts acylation of anisole with acetyl chloride affords a binary product with a total mass of 1.91 grams. This product is analyzed by gas chromatograph. The chromatogram shows two product peaks identified as ortho-methoxy acetophenone and para-methoxy acetophenone. Answer the following questions. H3CO Anisole MW: 108.14 H3C Acetyl chloride MW: 78.50 AICI3, THF 0 °C to rt, 4 h H3C H3CO + ortho-methoxy acetophenone MW: 150.17 GC uncorrected area = 12 mm² Response factor = 1.08 H₂CO CH3 Para-methoxy acetophenone MW: 150.17 GC uncorrected area = 134 mm² Response factor = 1.02 A) Using all the data given above calculate the molar percent compositions of each of the two isomeric products obtained in the reaction. Also, calculate the mass of each of the two isomeric products in grams. Show all work. B) A student conducted the above reaction with 2.64 g of anisole and 2.36 g of acetyl chloride. He purified the product mixture by column chromatography and obtained 0.1254 g of ortho-methoxy acetophenone and 1.6843 g of para- methoxy acetophenone. Show how you would find out the limiting reagent. Show how you would calculate the number of moles of the materials needed. Calculate the theoretical yield and percent yield for each of the isomeric products. Show all calculations and write down all appropriate formulas. A Friedel-Crafts acylation of anisole with acetyl chloride affords a binary product with a total mass of 1.91 grams. This product is analyzed by gas chromatograph. The chromatogram shows two product peaks identified as ortho-methoxy acetophenone and para-methoxy acetophenone. Answer the following questions. H3CO Anisole MW: 108.14 H3C Acetyl chloride MW: 78.50 AICI3, THF 0 °C to rt, 4 h H3C H3CO + ortho-methoxy acetophenone MW: 150.17 GC uncorrected area = 12 mm² Response factor = 1.08 H₂CO CH3 Para-methoxy acetophenone MW: 150.17 GC uncorrected area = 134 mm² Response factor = 1.02 A) Using all the data given above calculate the molar percent compositions of each of the two isomeric products obtained in the reaction. Also, calculate the mass of each of the two isomeric products in grams. Show all work. B) A student conducted the above reaction with 2.64 g of anisole and 2.36 g of acetyl chloride. He purified the product mixture by column chromatography and obtained 0.1254 g of ortho-methoxy acetophenone and 1.6843 g of para- methoxy acetophenone. Show how you would find out the limiting reagent. Show how you would calculate the number of moles of the materials needed. Calculate the theoretical yield and percent yield for each of the isomeric products. Show all calculations and write down all appropriate formulas.
Expert Answer:
Answer rating: 100% (QA)
2 A Given total mass or the product area 12 mm Para pordnet area a 134 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Factor A Factor B 1 2 3 4 Xj for Factor B 1 2 3 4 1 2500 2 9 10 6 9 8500 3 14 11 16 12 13250 Xi for Factor A 8333 8000 9000 7333 X8083
-
The results taken from a mass spectrum of chlorine gas show peaks at m/z 35.00 and m/z 37.00 (The m/z peaks on a mass spectrum identify the different isotopes of an element that are present in the...
-
A rectangular aluminum bar 12 mm thick and 50 mm wide is welded to fixed supports at the ends, and the bar supports a load W = 3.5 kN, acting through a pin as shown. Find the reactions at the...
-
1-Calculate amount of secondary compression in terms of inches 10 years following end of the primary consolidation. S.sc ang (12)= Coefficient of secondary compression=0.035 Thickness of clay layer-...
-
Captain's Cereal is considering introducing a variation of its current breakfast cereal, Crunch Stuff. This new cereal will be similar to the old, with the exception that it will contain sugar-coated...
-
The object provides methods for the creation of Statement objects that will be used to execute Oracle statements in the next step. a. Statement b. Connection c. DriverManager d. Driver
-
You are a small manufacturer of specialty medical devices located in Chicago, Illinois, and the market for your product is cardiologists in the United States. Does globalization affect your business?...
-
If an agency makes a loan at a below-market rate, what would be the nature of any expense recognized at the time of the loan? If it guaranteed a loan made by others, what would be the nature of any...
-
A string of length 0.650 m and a linear density of 2.02 x 10-3 kg/m is vibrating in the fundamental harmonic mode even as the tension is increased over time. How many oscillations does the string go...
-
Suppose Ann is working on a project with John. Both must decide whether to put into a decent amount of effort into the project. Since Ann is the leader of the project, her contribution determines...
-
Michael Smith and Joan have been dating for two years. Michael earns $6,000 monthly as the General manager of Siennas Health and Fitness store. Joan is a single mother currently working as a...
-
The 1964 Civil Rights Act prevents merchants, public accommodations, and employers from denying service and practicing discrimination in employment based on defined criteria. Develop an argument...
-
Describe how this scene illustrates the role of reflected appraisal and social comparison in forming a person's self-concept. Discuss how Ben and Katie could use the textbook material on Emotions to...
-
Please rewrite this letter with any and all punctuation you feel is necessary and required. July 5 2022 Manager, Faculty Affairs ABC County College To whom it concerns I am writing to offer my...
-
From the book Actions of Disrupted: My Misadventure in the Start-Up Bubble , Lyons talks about "The Bozo Explosion" and how it impacts that kinds of people that end up working at HubSpot. These...
-
In the topic of Lab Rats: How Silicon Valley made work miserable for the rest of us Lyons talks about the downside of constant change in the organizational context, with plenty of examples to back up...
-
Identify the constitutional amendment being violated in the following hypothetical situations: Jeremy's boss threatens to fire him if he does not work on Saturday nights, even though that is when he...
-
Critical reading SAT scores are distributed as N(500, 100). a. Find the SAT score at the 75th percentile. b. Find the SAT score at the 25th percentile. c. Find the interquartile range for SAT scores....
-
As a method for the preparation of alkenes, a weakness in the acid-catalyzed dehydration of alcohols is that the initially formed alkene (or mixture of alkenes) sometimes isomerizes under the...
-
Select the combination of alkyl bromide and potassium alkoxide that would be the most effective in the syntheses of the following ethers: (a) CH3OC(CH3)3 (b) (c) (CH3)3CCH2OCH2CH3 OCH
-
Write chemical equations for the reaction that takes place between each of the following pairs of reactants: (a) 3-Ethyl-3-pentanol and hydrogen chloride (b) 1-Tetradecanol and hydrogen bromide
-
Two loudspeakers \(42.0 \mathrm{~m}\) apart and facing each other emit identical \(115 \mathrm{~Hz}\) sinusoidal sound waves in a room where the sound speed is \(345 \mathrm{~m} / \mathrm{s}\). Susan...
-
A child's train whistle replicates a classic conductor's whistle from the early 1900 s. This whistle has two open-open tubes that produce two different frequencies. When you hear these two different...
-
A student waiting at a stoplight notices that her turn signal, which has a period of \(0.85 \mathrm{~s}\), makes one blink exactly in sync with the turn signal of the car in front of her. The blinker...

Study smarter with the SolutionInn App


