Question: A gas absorption tower contains two feed streams ? Fl: a gas containing 33.0 mole% 02, 66% N2, and 1% SO 2 at 250L/s at
A gas absorption tower contains two feed streams ? Fl: a gas containing 33.0 mole% 02, 66% N2, and 1% SO 2 at 250L/s at 35?C, and 1.2 atm; and F2: liquid water flowing at a rate of 15.0 mol/s at 35?C, and 1.2 atm. There are two product streams coming out this gas absorption tower ? P1: a liquid stream containing most of the entering water and a small amount of dissolved SO 2 ; and P2: a gas stream containing all of the entering O 2 and N 2 , the balance of the entering SO 2 , and some water vapour. The two product streams are in equilibrium with each other at 35?C and 1.2 atm. The equilibrium distribution of SO: and H 2 O between those streams is described by two relations:
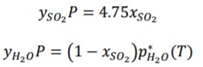
where x so2 , is the mole fraction of SO 2 : in the P1 liquid stream, y so2 , and yH 2 O are the mole fractions of SO 2 : and H 2 O in the P2 gas stream, respectively. P is the system pressure and p * H2O (T) is the vapour pressure of H 2 O at temperature T.
a. Draw and completely label a flow chart of this process, including component mole fractions in the labelling of all but the liquid water feed stream.
b. Show that the degrees of freedom for this process is zero by naming the unknown variables and the equations you can write to solve this problem.
c. Solve the simultaneous equations in part b to calculate the values of all the unknowns.
d. What is the percentage recovery of SO 2 ?
e. Comment percentage recovery the concentration of SO2 in the gas stream leaving the absorption column (consider that SO 2 is an air pollutant and has strict emissions standards). Propose methods for improving the recovery of SO2 and meeting the environmental regulations.
YsoP = 4.75xs02 Y0P = (1-XS0)PH0 (T)
Step by Step Solution
3.31 Rating (160 Votes )
There are 3 Steps involved in it
To tackle this problem lets go through each part stepbystep a Flow Chart For the flow chart we need to represent the input and output streams as follo... View full answer

Get step-by-step solutions from verified subject matter experts


