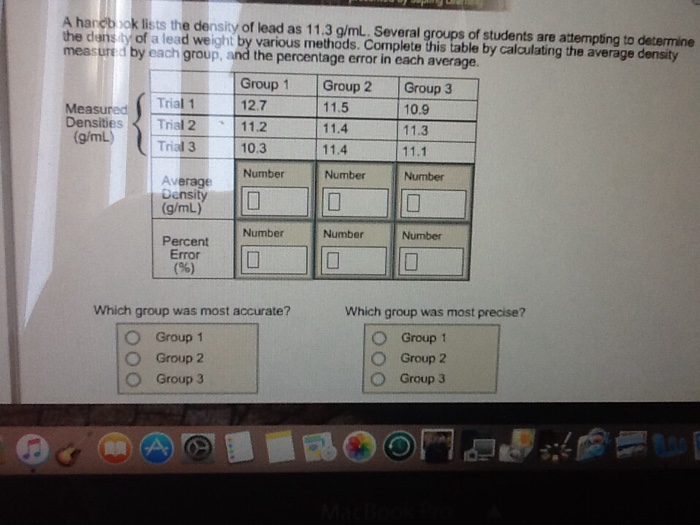
A handbook lists the density of lead as 11.3 g/mL. Several groups of students are attempting...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A handbook lists the density of lead as 11.3 g/mL. Several groups of students are attempting to determine the density of a lead weight by various methods. Complete this table by calculating the average density measured by each group, and the percentage error in each average. Measured Densities (g/mL) Trial 1 Trial 2 Trial 3 Average Density (g/mL) Percent Error (%) A Group 1 12.7 11.2 10.3 Number Which group was most accurate? Group 1 Group 2 Group 3 **** Number 0 Group 2 11.5 11.4 11.4 Number Number 0 Group 3 10.9 11.3 11.1 Number 0 Number 0 Which group was most precise? Group 1 Group 2 Group 3 A handbook lists the density of lead as 11.3 g/mL. Several groups of students are attempting to determine the density of a lead weight by various methods. Complete this table by calculating the average density measured by each group, and the percentage error in each average. Measured Densities (g/mL) Trial 1 Trial 2 Trial 3 Average Density (g/mL) Percent Error (%) A Group 1 12.7 11.2 10.3 Number Which group was most accurate? Group 1 Group 2 Group 3 **** Number 0 Group 2 11.5 11.4 11.4 Number Number 0 Group 3 10.9 11.3 11.1 Number 0 Number 0 Which group was most precise? Group 1 Group 2 Group 3
Expert Answer:
Answer rating: 100% (QA)
Group 1 Calculate the average density gmL as follows Density Trial 1Trial 2 Trial 3 Avera... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
The density of lead is 11.3 g/cm3. What is this value in kilograms per cubic meter?
-
The accountants for a city government are attempting to determine the appropriate method of reporting a series of unique financial transactions. They have reviewed all of the authoritative sources of...
-
The accountants for a city government are attempting to determine the appropriate method of reporting a series of unique financial transactions. They review all of the authoritative sources of United...
-
There is a bond on the spot-market. Price is 87.63 USD. Risk-free interest rate is 1.22%. The forward-price is 102.78 USD. Is there any arbitrage possibility if time to maturity is 9 months?
-
Briggs Excavation Company is planning an investment of $ 132,000 for a bulldozer. The bulldozer is expected to operate for 1,500 hours per year for five years. Customers will be charged $ 110 per...
-
Tab Corporation decided to establish Collon Company as a wholly owned subsidiary by transferring some of its existing assets and liabilities to the new entity. In exchange, Collon issued Tab 30,000...
-
CGI Federal, Inc., is a corporation that provides a number of services to the United States Passport Agency, included the processing of passport application. Passport applicants must submit sensitive...
-
Pixel Studio, Inc., is a small company that creates computer-generated animations for films and television. Much of the companys work consists of short commercials for television, but the company...
-
Based on the Lease Obligations by Year visualization, do the results make sense? Select answer from the options below No. There is a decreasing trend in finance lease and operating lease obligations,...
-
assume the underlying asset is a stock worth $55 today. Also assume the following spot prices will be realized in the future S 1 =$62 and S 2 =47. Also assume that C(50,1)=$7 and P(50,2)=$3. r=15%....
-
A source, approaching with speed u towards the open end of a stationary pipe of length L, is emitting a sound of frequency f. The farther end of the pipe is closed. The speed of sound in air is v and...
-
A 57 kg person on a merry-go-round is traveling in a circle with a radius of 2 m at a speed of 5 m/s. (a) What acceleration does the person experience? 12.5 m/s (b) What is the net horizontal force?...
-
When Wilson died on March 31, he had been retired for three years. What will be the due date for his final tax return?
-
Shirley borrowed $25,000 from her policy two years ago when her policy's adjusted cost base (ACB) was $10,000. This year, she repaid $20,000 of that loan. What amount can Shirley deduct from her...
-
Discuss with peers how each sees Implicit Bias negatively affecting group decision making? Give examples from your business/practice. Further discuss potential solutions to improve group decision...
-
If the remediation of an oil leak causes the Kochanek family to be displaced from their home for several months, what type of cost of contamination is incurred?
-
Pratt's Pizza Inc. is a small chain of take-out pizza restaurants. The average selling price of a Pratt's pizza is $15. Pratt's estimated sales for the first five months of the 2020 fiscal year are...
-
Complete the following acid-base reactions: (a) HCCH + NaH
-
Based on Lewis structures, predict the ordering of N-O bond lengths in NO+, NO2- and NO3-.
-
Using only the periodic table as your guide, select the most electronegative atom in each of the following sets: (a) Na, Mg, K, Ca; (b) P, S, As, Se; (c) Be, B, C, Si; (d) Zn, Ge, Ga, As.
-
A system goes from state 1 to state 2 and back to state 1. (a) What is the relationship between the value of E for going from state 1 to state 2 to that for going from state 2 back to state 1? (b)...
-
a. Find the rank of the given matrix. b. Confirm the result of (a) in MATLAB. 3 5 ST TNT 2 A=1 7
-
Prove that the product of two symmetric matrices is not necessarily symmetric.
-
BAw Perform the indicated operations, if defined, for the following vectors and matrices. -2 1 -3 1 1 A = 1 -3 2 1 32 B = V= W = 0 0 4 5

Study smarter with the SolutionInn App


