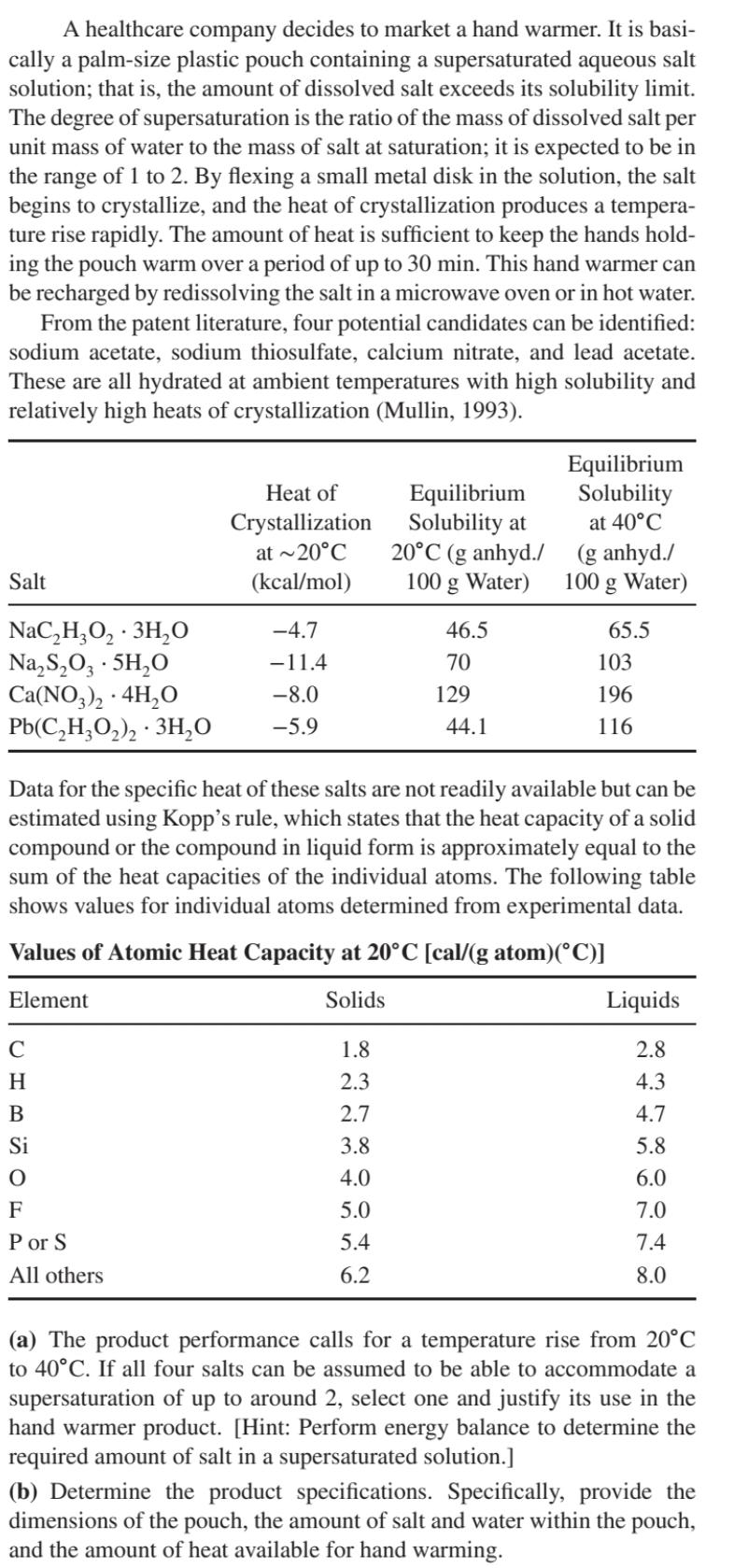
A healthcare company decides to market a hand warmer. It is basi- cally a palm-size plastic...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A healthcare company decides to market a hand warmer. It is basi- cally a palm-size plastic pouch containing a supersaturated aqueous salt solution; that is, the amount of dissolved salt exceeds its solubility limit. The degree of supersaturation is the ratio of the mass of dissolved salt per unit mass of water to the mass of salt at saturation; it is expected to be in the range of 1 to 2. By flexing a small metal disk in the solution, the salt begins to crystallize, and the heat of crystallization produces a tempera- ture rise rapidly. The amount of heat is sufficient to keep the hands hold- ing the pouch warm over a period of up to 30 min. This hand warmer can be recharged by redissolving the salt in a microwave oven or in hot water. From the patent literature, four potential candidates can be identified: sodium acetate, sodium thiosulfate, calcium nitrate, and lead acetate. These are all hydrated at ambient temperatures with high solubility and relatively high heats of crystallization (Mullin, 1993). Heat of Crystallization Equilibrium Equilibrium Solubility Solubility at at 40C Salt at ~20C (kcal/mol) 20C (g anhyd./ (g anhyd./ 100 g Water) 100 g Water) NaCHO 3HO NaSO, 5HO -4.7 46.5 65.5 -11.4 70 103 Ca(NO3)2 + 4HO . -8.0 129 196 Pb(C2H3O2)2.3HO -5.9 44.1 116 Data for the specific heat of these salts are not readily available but can be estimated using Kopp's rule, which states that the heat capacity of a solid compound or the compound in liquid form is approximately equal to the sum of the heat capacities of the individual atoms. The following table shows values for individual atoms determined from experimental data. Values of Atomic Heat Capacity at 20C [cal/(g atom)(C)] Element C Solids Liquids 1.8 2.8 H 2.3 4.3 B 2.7 4.7 Si 3.8 5.8 4.0 6.0 F Por S All others 5.0 7.0 5.4 7.4 6.2 8.0 (a) The product performance calls for a temperature rise from 20C to 40C. If all four salts can be assumed to be able to accommodate a supersaturation of up to around 2, select one and justify its use in the hand warmer product. [Hint: Perform energy balance to determine the required amount of salt in a supersaturated solution.] (b) Determine the product specifications. Specifically, provide the dimensions of the pouch, the amount of salt and water within the pouch, and the amount of heat available for hand warming. A healthcare company decides to market a hand warmer. It is basi- cally a palm-size plastic pouch containing a supersaturated aqueous salt solution; that is, the amount of dissolved salt exceeds its solubility limit. The degree of supersaturation is the ratio of the mass of dissolved salt per unit mass of water to the mass of salt at saturation; it is expected to be in the range of 1 to 2. By flexing a small metal disk in the solution, the salt begins to crystallize, and the heat of crystallization produces a tempera- ture rise rapidly. The amount of heat is sufficient to keep the hands hold- ing the pouch warm over a period of up to 30 min. This hand warmer can be recharged by redissolving the salt in a microwave oven or in hot water. From the patent literature, four potential candidates can be identified: sodium acetate, sodium thiosulfate, calcium nitrate, and lead acetate. These are all hydrated at ambient temperatures with high solubility and relatively high heats of crystallization (Mullin, 1993). Heat of Crystallization Equilibrium Equilibrium Solubility Solubility at at 40C Salt at ~20C (kcal/mol) 20C (g anhyd./ (g anhyd./ 100 g Water) 100 g Water) NaCHO 3HO NaSO, 5HO -4.7 46.5 65.5 -11.4 70 103 Ca(NO3)2 + 4HO . -8.0 129 196 Pb(C2H3O2)2.3HO -5.9 44.1 116 Data for the specific heat of these salts are not readily available but can be estimated using Kopp's rule, which states that the heat capacity of a solid compound or the compound in liquid form is approximately equal to the sum of the heat capacities of the individual atoms. The following table shows values for individual atoms determined from experimental data. Values of Atomic Heat Capacity at 20C [cal/(g atom)(C)] Element C Solids Liquids 1.8 2.8 H 2.3 4.3 B 2.7 4.7 Si 3.8 5.8 4.0 6.0 F Por S All others 5.0 7.0 5.4 7.4 6.2 8.0 (a) The product performance calls for a temperature rise from 20C to 40C. If all four salts can be assumed to be able to accommodate a supersaturation of up to around 2, select one and justify its use in the hand warmer product. [Hint: Perform energy balance to determine the required amount of salt in a supersaturated solution.] (b) Determine the product specifications. Specifically, provide the dimensions of the pouch, the amount of salt and water within the pouch, and the amount of heat available for hand warming.
Expert Answer:

Related Book For 

Accounting What the Numbers Mean
ISBN: 978-0078025297
10th edition
Authors: David H. Marshall, Wayne W. McManus, Daniel F. Viele
Posted Date:
Students also viewed these chemical engineering questions
-
I need help on this read from bottom to top. Image transcription text 7"} The second ?nancial statement to prepare is the statement of retained earnings. To determine the ending balance of...
-
___ raise capital for an infant corporation by making subscription agreements with subscribers in which the subscribers agree to purchase stock in the new corporation. Multiple choice question....
-
Ginter Co. holds Kolar Inc.s $10,000, 120-day, 9% note. The entry made by Ginter Co. when the note is collected, assuming no interest has been previously accrued, is: (a) Cash Notes Receivable (b)...
-
Periodically, the Hillsborough County (Florida) Water Department tests the drinking water of homeowners for contaminants such as lead and copper. The lead and copper levels in water specimens...
-
Determine whether each statement is true or false, and explain why. The line y = -2x + 5 intersects the point (3, -1).
-
Use the implicit finite difference method to solve the heat conduction problem on the unit square: \[\begin{aligned}& \frac{\partial^{2} u}{\partial x^{2}}=\frac{\partial u}{\partial t} \\& u(x, 0)=x...
-
The following is information for three local auto dealers: a. Using the information given in the above table, construct income statements for each company and the industry average. Assume that each...
-
Crane Co. has zero units of beginning work in process. During the period, 15,120 units were completed and transferred out, and there were 630 units of ending work in process one-fifth complete as to...
-
You are the Project Manager on the design team for a new bridge across the North Saskatchewan River. Your team consists of an Architectural Team, Structural Engineering Team, and an Electrical...
-
Add. -7 + 6y 61,3 3 812 Simplify your answer as much as possible.
-
Solve the inequality. (Enter your answer using interval notation.) 4x3x2 < 6 - 24x (-00,00) X
-
Burns Company reported $931.480 million in net income in 2021. On January 1, 2021, the company had 404 million shares of common stock outstanding. On March 1, 2021, 26.4 million new shares of common...
-
Find the slope of the line that goes through the given points. (5, 7) and (7,3)
-
Solve this system of equations -3x-2y - 1x + 2y = 12 4
-
Describe the VAR approach to market risk. Is VAR related to the normal distribution? Can VAR handle extreme events (black swans)? Explain how VAR is applied to portfolios.
-
Three forces with magnitudes of 70pounds, 40 pounds, and 60 pounds act on an object at angles of 30, 45, and 135, respectively, with the positive x-axis. Find the direction and magnitude of the...
-
For a chordwise flexible airfoil obtain the quasi unsteady edge velocity, Eq. 8.41, and the suction force coefficient, Eq. 8.42, formulae assuming that the parabolic camber of the airfoil, whose...
-
Derive the equations of continuity, Eq. 8.44, and the vorticity transport, Eq. 8.45, for skewed coordinates as shown in Fig. 8.38. Eq. 8.44 Eq. 8.45 Fig. 8.38 de = tan -[-(h+WaLE) /U]
-
Obtain the quasi steady lift coefficient, Eq. 8.51, and the boundary layer edge velocity, Eq. 8.52 for a chordwise flexible and parabolically cambered thin airfoil whose equation is given by Eq....

Study smarter with the SolutionInn App


