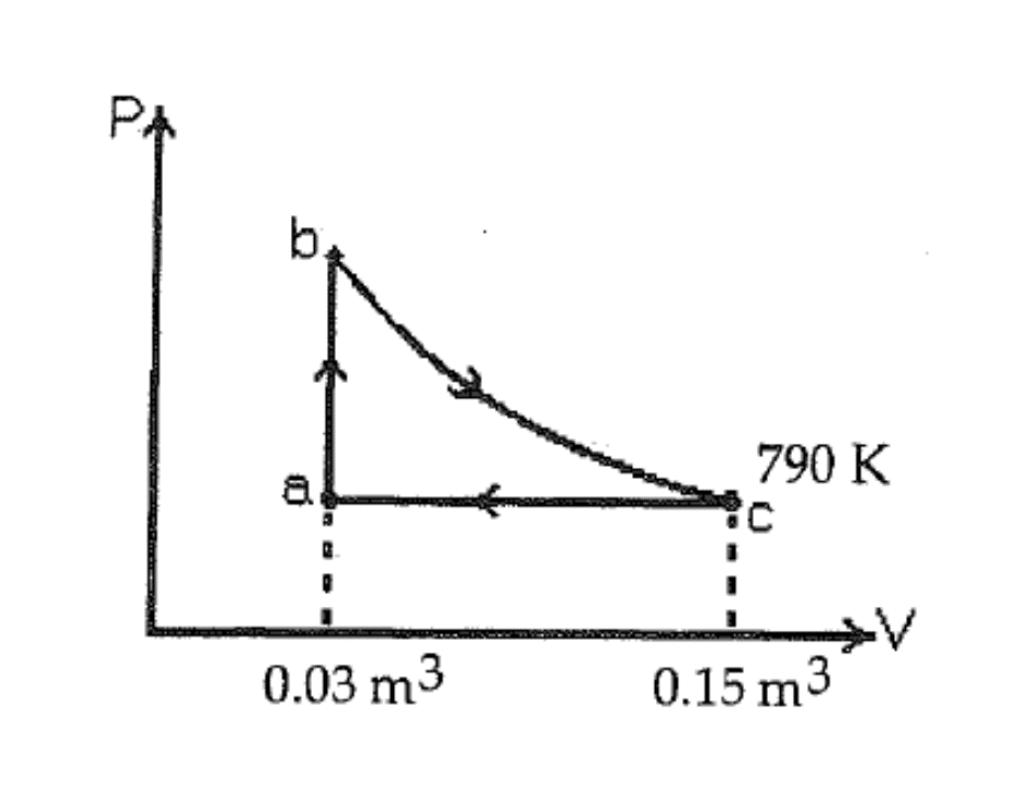
A heat engine takes 6.0 moles of an ideal gas through the reversible cycle abca , on
Fantastic news! We've Found the answer you've been seeking!
Question:
A heat engine takes 6.0 moles of an ideal gas through the reversible cycle abca, on the pV diagram, as shown. The path bc is an isothermal process. The temperature at c is 790 K, and the volumes at a and c are 0.03 m3 and 0.15 m3 , respectively. The molar heat capacity at constant volume, of the gas, is 26 J/mol K. In this figure, for the path bc, the work done by the gas, in kJ is?
Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Posted Date:




