(a) Identify the bond that is broken in ATP hydrolysis to ADP. Select the bond by...
Fantastic news! We've Found the answer you've been seeking!
Question:
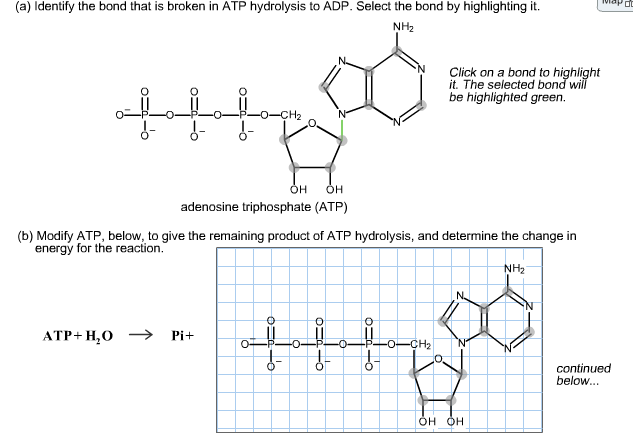
Transcribed Image Text:
(a) Identify the bond that is broken in ATP hydrolysis to ADP. Select the bond by highlighting it. NH₂ OH OH adenosine triphosphate (ATP) (b) Modify ATP, below, to give the remaining product of ATP hydrolysis, and determine the change in energy for the reaction. ATP+H₂O →>> Pi+ O ↓- O !- !- Click on a bond to highlight it. The selected bond will be highlighted green. O -CH₂ OH OH NH₂ continued below... (a) Identify the bond that is broken in ATP hydrolysis to ADP. Select the bond by highlighting it. NH₂ OH OH adenosine triphosphate (ATP) (b) Modify ATP, below, to give the remaining product of ATP hydrolysis, and determine the change in energy for the reaction. ATP+H₂O →>> Pi+ O ↓- O !- !- Click on a bond to highlight it. The selected bond will be highlighted green. O -CH₂ OH OH NH₂ continued below...
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below Answer Ans A AT... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these accounting questions
-
Adenosine triphosphate (ATP) is often referred to as a biological energy source. What does this mean? Explain how ATP is used in biological organisms to produce proteins from amino acids.
-
The free energy of hydrolysis of an ((1(4) glycosidic bond is - 15.5 kJ ( mol-1, whereas that of an ((1(6) glycosidic bond is -7.1 kJ ( mol-1. Use these data to explain why glycogen debranching...
-
Give examples of a dehydration synthesis reaction and a hydrolysis reaction.
-
The IQ and VIQ are tools insurance producers use to learn more about internal assumptions regarding factors that ultimately control policy performance. They have information in all of the following...
-
Who pays Social Security taxes: the employer, the employee, or both? How is the deduction for Social Security and Medicare (FICA) computed?
-
If you put up $45,000 today in exchange for a 6.25 percent, 15-year annuity, what will the annual cash flow be?
-
Use information from Section 6.7 to estimate which form of electromagnetic radiation is the lowest energy ionizing radiation. Data from section 6.7 When we first introduced the concept of the...
-
Logan Bruno Company has just received the August 31, 2008, bank statement, which is summarized below. The general ledger Cash account contained the following entries for the month of August. Deposits...
-
If an employee's benefit is paid from a qualified plan in the form of a qualifying lump-sum benefit at retirement, the employee's cost basis will be recovered _______________ with an income tax...
-
Below is the Trial balance of Miss Piggy & Kermit Inc. after his first years trading: Miss Piggy & Kermit Inc Trial Balance as of 30 June 20X8 Dr. Cr $ $ Revenue 99,082 Purchases 71,409 Rent...
-
If a company hires Nielsen to gather information of a very specific problem that a company encounters, Nielsen likely collects Causal O Primary O Descriptive O Exploratory O Secondary data from...
-
There are two accounting principles that are used to determine the net result of the project s work, whether profit or loss, for a specific financial period. These two foundations are: the accrual...
-
What are some key performance indicators (KPIs) used in the accounting and financial analysis of a manufacturing company?
-
Products in finished goods inventory that are normally kept on hand in order to quickly fill customer orders instead of being made for a specific customer order. What is the role of an accountant in...
-
The following is/are a disadvantage(s) of buying rather than making a component of a company's product: quality control specifications may not be met the outside supplier could increase prices...
-
Match the words with the term Question 1 options: semi-variable costs difference between revenue and total costs variable costs non-cash expense How are manufacturing overhead costs allocated and...
-
It is now April 1, and you are considering investing in 90 day bank bills in two months' time (or 60 days). That is, on June 1 you intend to purchase bank bills with a face value of $2,000,000 at...
-
X-1 Find the domain of the function f(x) : x 1 2 - O (-00, -1) U (-1, ) O (-00, 1) U (1, ) O -00, -1) U (-1, 1) U (1, 0) O (- 1, 1)
-
Name or write the condensed structural formula for the following compounds: (a) 4-methyl-2-pentene (b) cis-2,5-dimethyl-3-hexene (c) ortho-dimethylbenzene (d) HC'CCH2CH3 (e) trans-CH3CH"CHCH2CH2CH2CH3
-
At room temperature and pressure RbI crystallizes with the NaCl-type structure. (a) Use ionic radii to predict the length of the cubic unit cell edge. (b) Use this value to estimate the density. (c)...
-
The [Ni (H2O)6]2+ ion has an absorption maximum at about 725 nm, whereas the [Ni (H2O)6]2+ ion absorbs at about 570nm. Predict the color of a solution of each ion. (b) The [Ni(en)3]2+ ion occurs at...
-
A typical timber wolf has a mass of \(40 \mathrm{~kg}\), a typical jackrabbit a mass of \(2.5 \mathrm{~kg}\). Given the scaling law presented in the passage, we'd expect the specific metabolic rate...
-
A standard gold bar stored at Fort Knox, Kentucky, is 7.00 inches long, 3.63 inches wide, and 1.75 inches tall. Gold has a density of \(19,300 \mathrm{~kg} / \mathrm{m}^{3}\). What is the mass of...
-
A typical timber wolf has a mass of \(40 \mathrm{~kg}\), a typical jackrabbit a mass of \(2.5 \mathrm{~kg}\). Given the scaling law presented in the passage, we'd expect the wolf to use times more...

Study smarter with the SolutionInn App


