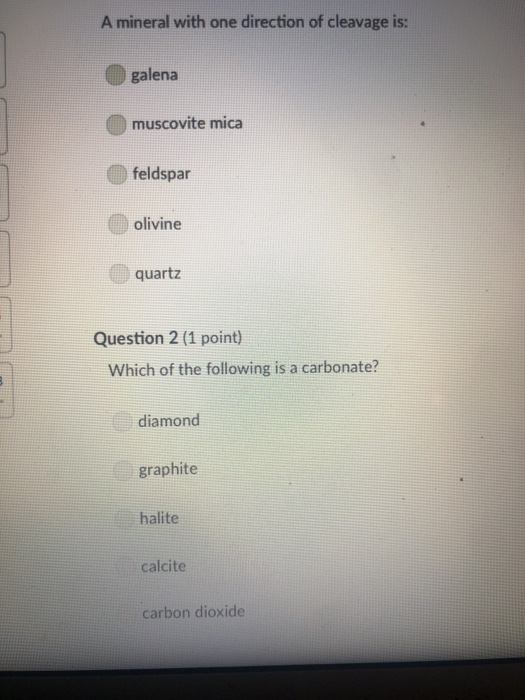
A mineral with one direction of cleavage is: galena muscovite mica feldspar olivine quartz Question 2...
Fantastic news! We've Found the answer you've been seeking!
Question:
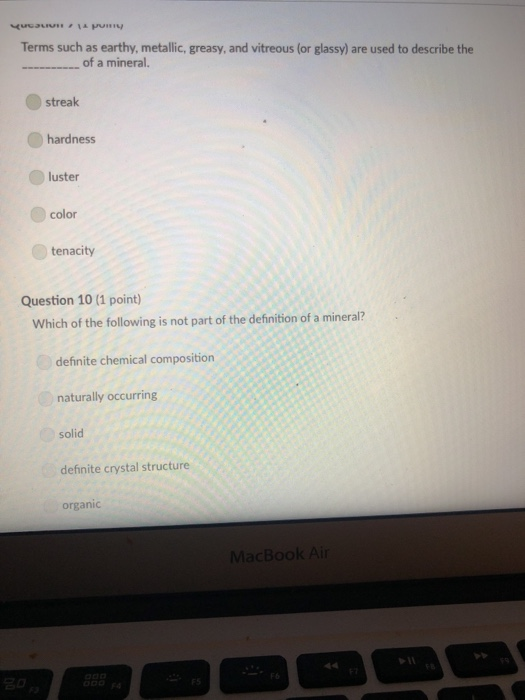
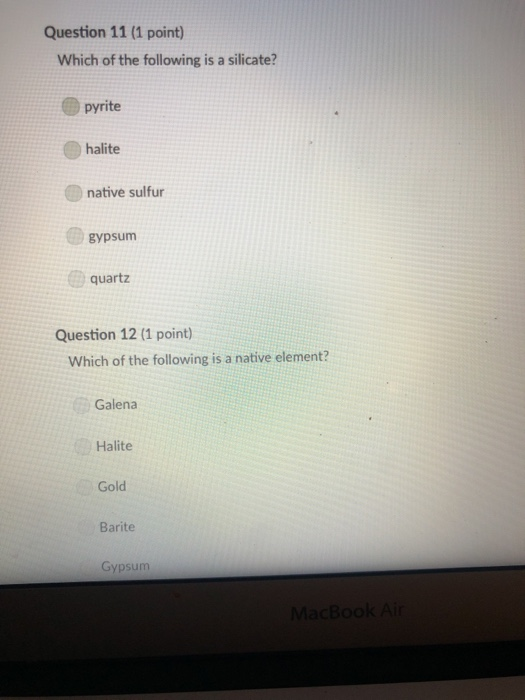
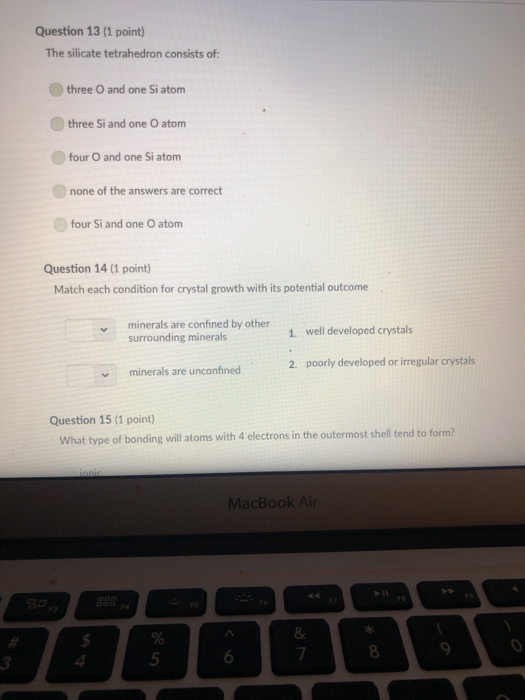
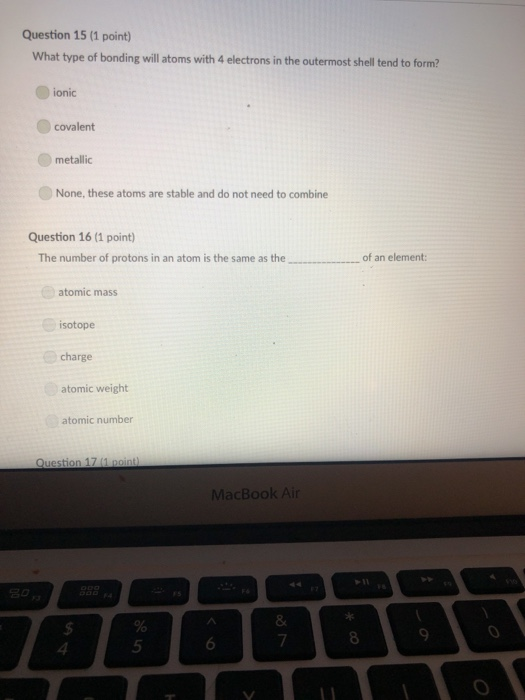
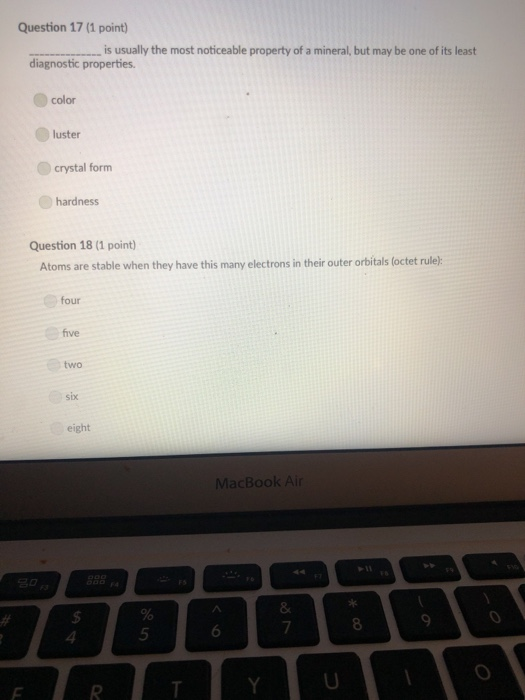
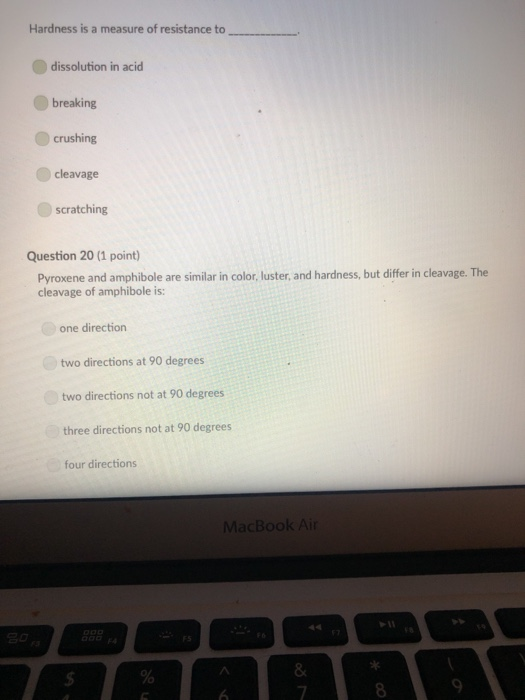
Transcribed Image Text:
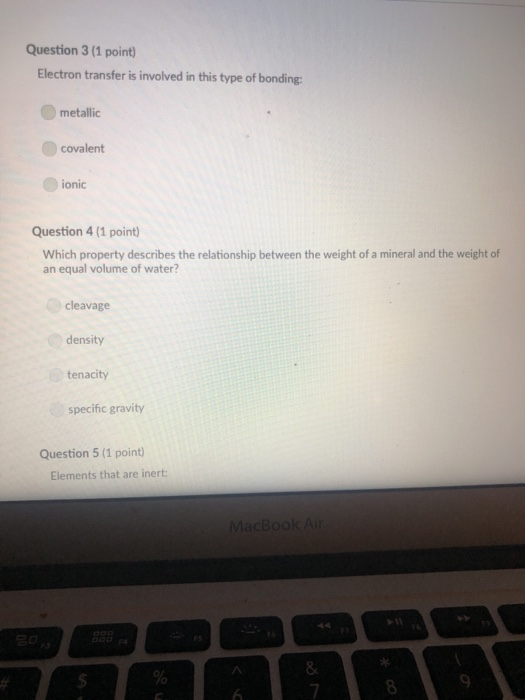
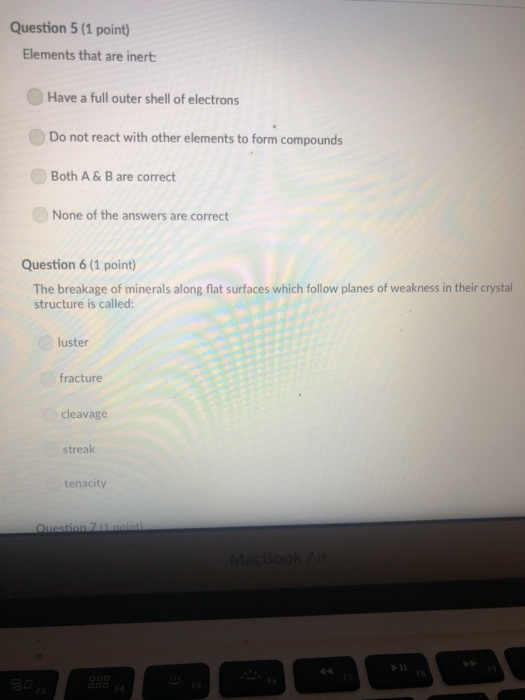
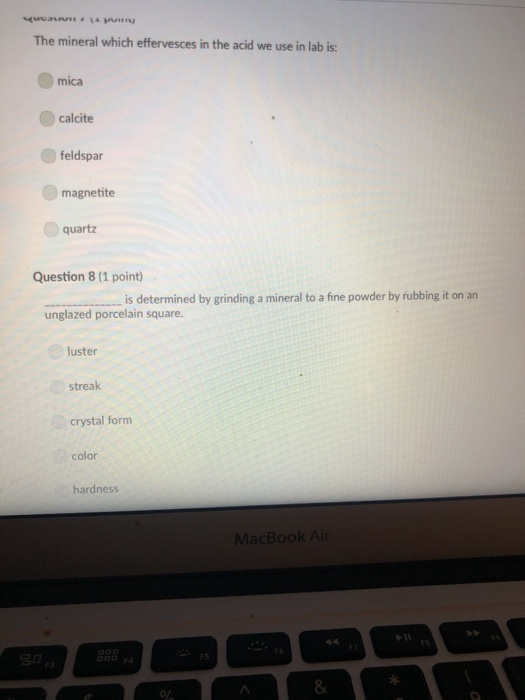
A mineral with one direction of cleavage is: galena muscovite mica feldspar olivine quartz Question 2 (1 point) Which of the following is a carbonate? diamond graphite halite calcite carbon dioxide Question 3 (1 point) Electron transfer is involved in this type of bonding: metallic covalent ionic Question 4 (1 point) Which property describes the relationship between the weight of a mineral and the weight of an equal volume of water? cleavage density tenacity specific gravity Question 5 (1 point) Elements that are inert: $ 000 % MacBook Air & > 11 8 Question 5 (1 point) Elements that are inert: Have a full outer shell of electrons Do not react with other elements to form compounds Both A & B are correct None of the answers are correct Question 6 (1 point) The breakage of minerals along flat surfaces which follow planes of weakness in their crystal structure is called: luster fracture cleavage streak tenacity Question 711 point) F4 F5 MacBook Air F6 un proy The mineral which effervesces in the acid we use in lab is: mica calcite feldspar magnetite quartz Question 8 (1 point) unglazed porcelain square. luster streak is determined by grinding a mineral to a fine powder by rubbing it on an crystal form color hardness F5 MacBook Air F6 & 15 E Stations pro Terms such as earthy, metallic, greasy, and vitreous (or glassy) are used to describe the of a mineral. streak 20 hardness luster color tenacity Question 10 (1 point) Which of the following is not part of the definition of a mineral? definite chemical composition naturally occurring solid definite crystal structure organic F5 MacBook Air Question 11 (1 point) Which of the following is a silicate? pyrite halite native sulfur gypsum quartz Question 12 (1 point) Which of the following is a native element? Galena Halite Gold Barite Gypsum MacBook Air 3 Question 13 (1 point) The silicate tetrahedron consists of: three O and one Si atom 80 three Si and one O atom four O and one Si atom none of the answers are correct four Si and one O atom Question 14 (1 point) Match each condition for crystal growth with its potential outcome ionic minerals are confined by other surrounding minerals 4 minerals are unconfined Question 15 (1 point) What type of bonding will atoms with 4 electrons in the outermost shell tend to form? % 5 FS 1. well developed crystals * 2. poorly developed or irregular crystals 6 MacBook Air & 7 ∞ * 8 9 Question 15 (1 point) What type of bonding will atoms with 4 electrons in the outermost shell tend to form? ionic 20 covalent metallic None, these atoms are stable and do not need to combine Question 16 (1 point) The number of protons in an atom is the same as the atomic mass isotope charge atomic weight atomic number Question 17 (1 point) 4 999 % 5 MacBook Air 6 & 7 of an element: ➤11 8 0 # Question 17 (1 point) F diagnostic properties. color F3 luster crystal form hardness Question 18 (1 point) Atoms are stable when they have this many electrons in their outer orbitals (octet rule): four five two six is usually the most noticeable property of a mineral, but may be one of its least eight $ 4 R % 5 T MacBook Air 6 Y & 7 U *00 8 1 Hardness is a measure of resistance to dissolution in acid 20 breaking crushing cleavage scratching Question 20 (1 point). Pyroxene and amphibole are similar in color, luster, and hardness, but differ in cleavage. The cleavage of amphibole is: one direction two directions at 90 degrees two directions not at 90 degrees three directions not at 90 degrees four directions $ F4 % 5 F5 MacBook Air F6 & 7 * 8 A mineral with one direction of cleavage is: galena muscovite mica feldspar olivine quartz Question 2 (1 point) Which of the following is a carbonate? diamond graphite halite calcite carbon dioxide Question 3 (1 point) Electron transfer is involved in this type of bonding: metallic covalent ionic Question 4 (1 point) Which property describes the relationship between the weight of a mineral and the weight of an equal volume of water? cleavage density tenacity specific gravity Question 5 (1 point) Elements that are inert: $ 000 % MacBook Air & > 11 8 Question 5 (1 point) Elements that are inert: Have a full outer shell of electrons Do not react with other elements to form compounds Both A & B are correct None of the answers are correct Question 6 (1 point) The breakage of minerals along flat surfaces which follow planes of weakness in their crystal structure is called: luster fracture cleavage streak tenacity Question 711 point) F4 F5 MacBook Air F6 un proy The mineral which effervesces in the acid we use in lab is: mica calcite feldspar magnetite quartz Question 8 (1 point) unglazed porcelain square. luster streak is determined by grinding a mineral to a fine powder by rubbing it on an crystal form color hardness F5 MacBook Air F6 & 15 E Stations pro Terms such as earthy, metallic, greasy, and vitreous (or glassy) are used to describe the of a mineral. streak 20 hardness luster color tenacity Question 10 (1 point) Which of the following is not part of the definition of a mineral? definite chemical composition naturally occurring solid definite crystal structure organic F5 MacBook Air Question 11 (1 point) Which of the following is a silicate? pyrite halite native sulfur gypsum quartz Question 12 (1 point) Which of the following is a native element? Galena Halite Gold Barite Gypsum MacBook Air 3 Question 13 (1 point) The silicate tetrahedron consists of: three O and one Si atom 80 three Si and one O atom four O and one Si atom none of the answers are correct four Si and one O atom Question 14 (1 point) Match each condition for crystal growth with its potential outcome ionic minerals are confined by other surrounding minerals 4 minerals are unconfined Question 15 (1 point) What type of bonding will atoms with 4 electrons in the outermost shell tend to form? % 5 FS 1. well developed crystals * 2. poorly developed or irregular crystals 6 MacBook Air & 7 ∞ * 8 9 Question 15 (1 point) What type of bonding will atoms with 4 electrons in the outermost shell tend to form? ionic 20 covalent metallic None, these atoms are stable and do not need to combine Question 16 (1 point) The number of protons in an atom is the same as the atomic mass isotope charge atomic weight atomic number Question 17 (1 point) 4 999 % 5 MacBook Air 6 & 7 of an element: ➤11 8 0 # Question 17 (1 point) F diagnostic properties. color F3 luster crystal form hardness Question 18 (1 point) Atoms are stable when they have this many electrons in their outer orbitals (octet rule): four five two six is usually the most noticeable property of a mineral, but may be one of its least eight $ 4 R % 5 T MacBook Air 6 Y & 7 U *00 8 1 Hardness is a measure of resistance to dissolution in acid 20 breaking crushing cleavage scratching Question 20 (1 point). Pyroxene and amphibole are similar in color, luster, and hardness, but differ in cleavage. The cleavage of amphibole is: one direction two directions at 90 degrees two directions not at 90 degrees three directions not at 90 degrees four directions $ F4 % 5 F5 MacBook Air F6 & 7 * 8
Expert Answer:
Answer rating: 100% (QA)
1 correct answer is Muscovite mica Explanation Quartz and galena have 3 directions Feldspar and olivine have 2 directions of cleavage Muscovite mica has one direction of cleavage 2 correct answer is c... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these law questions
-
Diamond and graphite are both pure carbon. Yet diamond is the hardest natural substance and graphite is one of the softest. Why?
-
An Ionic Crystal Figure shows eight point charges arranged at the corners of a cube with sides of length d. The values of the charges are +q and -q, as shown. This is a model of one cell of a cubic...
-
Carbon dioxide is stripped from water by air in a wetted-wall tube. At a certain location, where the pressure is 10 atM and the temperature is 25oC, the mass-transfer flux of CO2 is 1.62 Ibmouh-ft2....
-
Mario and Kaitlin are married and file a joint tax return. They have adjusted gross income of $385,000 that includes $4,700 of investment income ($3,000 short-term capital gains and $1,700 of...
-
Explain how to perform a projected financial analysis.
-
An article in the Journal of Manufacturing Systems (1991, vol. 10, pp. 3240) described an experiment to investigate the effect of four factors, P = waterjet pressure, F = abrasive flow rate, G =...
-
During February, Peters Hardware made sales of \($38,600\) and ended the month with inventories totaling \($5,400.\) Cost of Goods Sold was \($23,200.\) Total operating expenses were \($8,700.\) How...
-
1. Table 1 can be used to obtain the demand curve for a monopolist who manufactures and sells a unique type of camera. The first column gives several production quantities in thousands of cameras,...
-
Astore pays $24 for an item and has a markup rate of 125%.What is the retail cost of the item? Retail prince is $
-
Which series has the highest beta. BraveNewCoin Liquid Index for Bitcoin 1D BNC Trading Brave Ne Yellow Green Blue Orange
-
Discuss Lewin's Change Management Model theories from writers proponents of those approaches from a process perspective and the other from a people perspective.
-
A wheel rotating with a constant angular acceleration turns through 22 revolutions dur- ing a 4 s time interval. Its angular velocity at the end of this interval is 12 rad/s. What is the angular...
-
1. For the reaction: A+ 3B 2C+ D, what is the order of the above reaction with respect to [B]? Initial Rate (M-s) [A] 0.023 0.054 0.054 [B] 0.048 0.048 0.019 (B) 1.5 (D) 3 2. Which plot shows a...
-
The car's tires have a radius of = 13" = 33 cm and the tires do not slip. 2. What is a correct setup to calculate the angular acceleration of the car tires
-
? Statement of owner's equity Brenda Tooley owns and operates Speedy Delivery Services. On January 1, 20Y7, Brenda Tooley, Capital had a balance of $879,500. During the year, Brenda invested an...
-
165. There is lymphoid tissue also located within the lining of the major tracts (respiratory, digestive and urogenital tracts) called mucosa- associated lymphoid tissue (MALT). It constitutes about...
-
2 (s)' Solid lead(II) hydroxide, Pb(OH), in a test tube will dissolve when excess 6 M NaOH is added. Solid iron(II) hydroxide, Fe(OH), however, does not dissolve when excess 6 M NaOH is added. Answer...
-
The cost curve for the city water supply is C(Q) = 16 + 1/4 Q2, where Q is the amount of water supplied and C(Q) is the cost of providing Q acre-feet of water. (An acre-foot is the amount of water...
-
Write the balanced molecular and net ionic equations for each of the following neutralization reactions: (a) Aqueous acetic acid is neutralized by aqueous barium hydroxide. (b) Solid chromium(III)...
-
The reaction between ethyl bromide (C2H5Br) and hydroxide ion in ethyl alcohol at 330 K, C2H5Br(alc) + OH (alc) -- C2H5OH(I) + Br(aIc), is first order each in ethyl bromide and hydroxide ion. When...
-
Consider the hypothetical molecule B-A == B. How could you use an experimentally determined structure of the molecule to decide whether resonance is important in it?
-
The following reaction takes place in a system consisting of \(3 \mathrm{~mol} \mathrm{CH}_{4}, 5 \mathrm{~mol} \mathrm{H}_{2} \mathrm{O}\), \(1 \mathrm{~mol} \mathrm{CO}\) and \(4 \mathrm{~mol}...
-
A system is said to be in thermodynamic equilibrium if its (a) Temperature remains unchanged (b) Pressure remains unchanged (c) Chemical potential remains unchanged (d) Temperature, pressure and...
-
A system initially containing \(3 \mathrm{~mol} \mathrm{CO}_{2}, 5 \mathrm{~mol} \mathrm{H}_{2}\) and \(1 \mathrm{~mol} \mathrm{H}_{2} \mathrm{O}\) is undergoing the following reactions: \[...

Study smarter with the SolutionInn App


