A mixture of two ideal gases X and Y has n, and n, in the ratio...
Fantastic news! We've Found the answer you've been seeking!
Question:
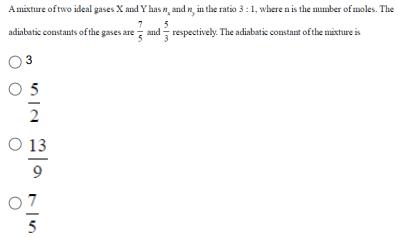
Transcribed Image Text:
A mixture of two ideal gases X and Y has n, and n, in the ratio 3:1, where n is the number of moles. The 7 5 adiabatic constants of the gases are t and respectively. The adiabatic constant of the mixture is 03 546555 O 13 A mixture of two ideal gases X and Y has n, and n, in the ratio 3:1, where n is the number of moles. The 7 5 adiabatic constants of the gases are t and respectively. The adiabatic constant of the mixture is 03 546555 O 13
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these physics questions
-
Given the following data, use least-squares regression to derive a trend equation. What is your estimate of the demand in period 7? In period 12? PERIOD DEMAND 1 7 2 9 m 5 4 11 5 10 6 13
-
Two gases X (molecular weight Mx) and Y (molecular weight My; My > Mx) are at the same temperature T in two different containers. Their root mean square velocities are Cx and Cy respectively. If the...
-
A container has a mixture of two gases: n1 mol of gas 1 having molar specific heat C1 and n2 mol of gas 2 of molar specific heat C2. (a) Find the molar specific heat of the mixture. (b) What If? What...
-
A test rocket is launched by accelerating it along a 200.0-m incline at 1.25 m/s2 starting from rest at point A (Figure 3.45.) The incline rises at 35.0o above the horizontal, and at the instant the...
-
Plaintiff owns a 150-acre tract of land in the town of Mamaroneck, which has been leased by a private country club since 1921. In 1922, the area where the country club was located became the subject...
-
An internal auditor determines that the process is not designed adequately to reduce the underlying risks to an acceptable level. Which of the following should the internal auditor do next? a. Write...
-
Describe common legal issues for behavioral health professionals.
-
Why can undue emphasis on labor efficiency variances lead to excess work in process inventories?
-
if V 3 = 1 0 s i n 1 0 0 t and V 4 = 9 s i n 1 0 0 0 t what is the current through the load resistor R 9 at t = 1 9 8 9 . 8 9 5 ? Resistor R 9 is a 1 1 6 watt carbon resistor, does it catch on fire...
-
Imagine a sedimentary basin on some planetary body that is confined on all sides and filled to a depth of 1 km. The horizontal strains, xx and yy are both zero, and the vertical stress at any depth...
-
10 Big data gets its name from the reality that about 90% of the world's data was created in the past two years-a fact that is changing the way companies view and manage data.. True False
-
In what circumstances can parents be held liable for the tortious acts of their children?
-
What is the difference between trespass to chattels and conversion, and what does a court consider when distinguishing between these two torts?
-
True Or False Under the retained control exception to the nonliability rule for independent contractors, an employer is considered to have retained control if the employer maintains control over the...
-
True Or False Some courts allow words alone to constitute an assault.
-
Under the doctrine of ____________ ____________, an individual is held liable for the tortious acts of another.
-
Your client, a real estate company, is evaluating a real estate project of $15,000,000 that will provide them with yearly NOI of $1,950,000 for the next 10 years and can then be resold for an amount...
-
At the beginning of the year, Lam Ltd. had total assets of $800,000 and total liabilities of $500,000. Use this information to answer each of the following independent questions. (a) If Lam's total...
-
From information in the Data section, calculate the standard Gibbs energy and the equilibrium constant at (a) 25C (b) 50C for the reaction CH4(g) + 3 C12(g) ='0CHC13(l) + 3 HCl(g). Assume that the...
-
A certain gas obeys the van der Waals equation with a =0.76 m6 Pa mol-2, its volume is found to be 4.00 X 10-4 m3 mol-1 at 288 K and 4.0 MPa. From this information calculate the van der Waals...
-
The isothermal compressibility of lead at 293 K is 2.21 X 10-6 atm-1, Calculate the pressure that must be applied in order to increase its density by 0.08 per cent.
-
A typical muscle fiber is 2.0 cm long and has a cross-section area of 3.1 10-9 m 2 . When the muscle fiber is stimulated, it pulls with a force of 1.2 mN. What is the work done by the muscle fiber...
-
You are pulling a child in a wagon. The rope handle is inclined upward at a 60 angle. The tension in the handle is 20 N. How much work do you do if you pull the wagon 100 m at a constant speed?
-
A wind turbine works by slowing the air that passes its blades and converting much of the extracted kinetic energy to electric energy. A large wind turbine has 45-m-radius blades. In typical...

Study smarter with the SolutionInn App



