A molecule or ion that donates the hydrogen in a hydrogen bond is a hydrogen bond...
Fantastic news! We've Found the answer you've been seeking!
Question:
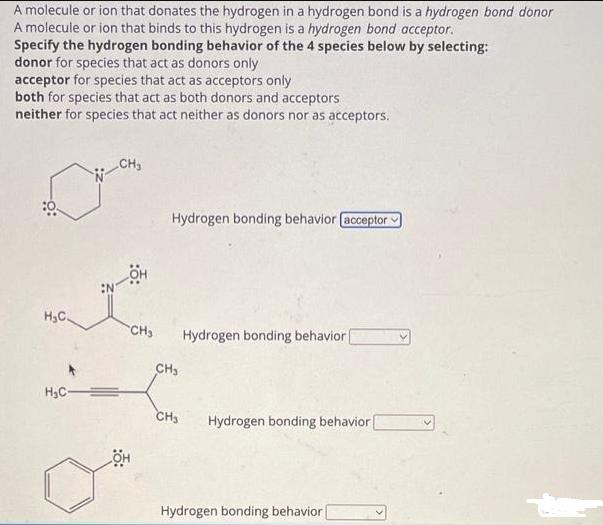
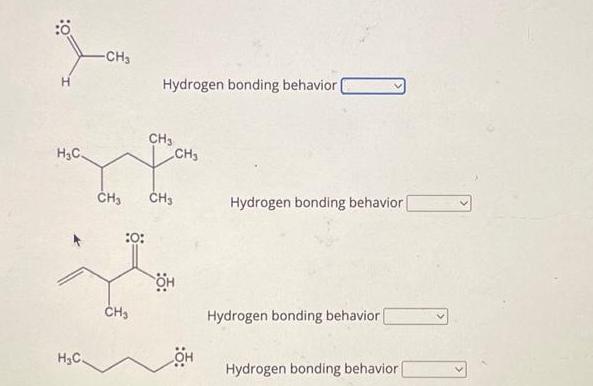
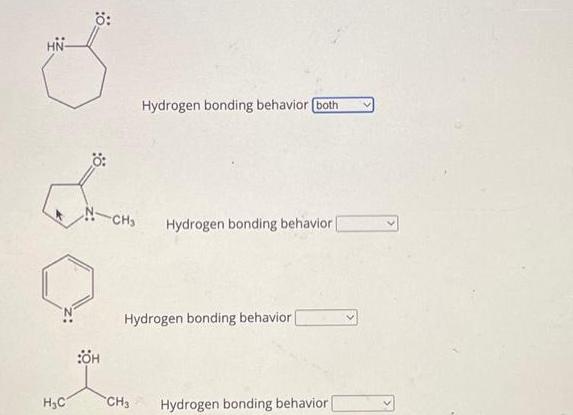
Transcribed Image Text:
A molecule or ion that donates the hydrogen in a hydrogen bond is a hydrogen bond donor A molecule or ion that binds to this hydrogen is a hydrogen bond acceptor. Specify the hydrogen bonding behavior of the 4 species below by selecting: donor for species that act as donors only acceptor for species that act as acceptors only both for species that act as both donors and acceptors neither for species that act neither as donors nor as acceptors. H₂C H₂C= CH₂ HO Hydrogen bonding behavior [acceptor CH₂ Hydrogen bonding behavior ( OH CH₂ CH₂ Hydrogen bonding behavior Hydrogen bonding behavior Ö: H₂C H₂C -CH3 :0: Hydrogen bonding behavior. CH3 CH₂ CH₂ CH3 он CH₂ OH Hydrogen bonding behavior Hydrogen bonding behavior Hydrogen bonding behavior | H₂C Ö: :ÖH -CH₂ Hydrogen bonding behavior (both Hydrogen bonding behavior | Hydrogen bonding behavior CH3 Hydrogen bonding behavior A molecule or ion that donates the hydrogen in a hydrogen bond is a hydrogen bond donor A molecule or ion that binds to this hydrogen is a hydrogen bond acceptor. Specify the hydrogen bonding behavior of the 4 species below by selecting: donor for species that act as donors only acceptor for species that act as acceptors only both for species that act as both donors and acceptors neither for species that act neither as donors nor as acceptors. H₂C H₂C= CH₂ HO Hydrogen bonding behavior [acceptor CH₂ Hydrogen bonding behavior ( OH CH₂ CH₂ Hydrogen bonding behavior Hydrogen bonding behavior Ö: H₂C H₂C -CH3 :0: Hydrogen bonding behavior. CH3 CH₂ CH₂ CH3 он CH₂ OH Hydrogen bonding behavior Hydrogen bonding behavior Hydrogen bonding behavior | H₂C Ö: :ÖH -CH₂ Hydrogen bonding behavior (both Hydrogen bonding behavior | Hydrogen bonding behavior CH3 Hydrogen bonding behavior
Expert Answer:
Answer rating: 100% (QA)
Hydrogen bond occurs usually the electronegative atom is oxygen nitrogen or fluorine which has a par... View the full answer

Related Book For 

Posted Date:
Students also viewed these psychology questions
-
Within the context of rapidly evolving regulatory landscapes and heightened ethical scrutiny, how do advanced organizations navigate the ethical complexities of technological innovation and...
-
What is the value of y[1] [2] ? int y[3][3]={{1,2,3},{4,5,6}, {7,8,9}};
-
On January 1, 2025, ABC Company, a calendar-year company, issued $2,000,000 of notes payable, of which $500,000 is due on January 1 for each of the next four years. What is the proper balance sheet...
-
1. Which type contains a single character enclosed within single quotes? A. Character B. Numeric C. Floating point 2. The modulus operator uses, B. - B. < A. + 3. Every variable should be separated...
-
What does it mean for an operating system to support multitasking? How is multitasking different from time sharing?
-
1. Daxkos David Gray says, Im a big believer in corporate culture as a competitive advantage. Do you agree? Explain. 2. Why do company cultures like the ones at New Belgium Brewing Company and Daxko...
-
A W14 \(\times 30\) structural A992 steel column is pin connected at is ends and has a length \(L=12 \mathrm{ft}\). Determine the maximum eccentric load \(P\) that can be applied so the column does...
-
Data concerning a recent periods activity in the Prep Department, the first processing department in a company that uses process costing, appear below: A total of 20,100 units were completed and...
-
Write a program for the following expression based on the 8086 arithmetic instructions to find X. C X=+(5E B) +[A * (F3 - B)] The content of register CL (C=CL) is 48, which store in the flag...
-
Raymond Constructions trial balance at December 31, 2020, is presented as follows. All 2020 transactions have been recorded except for the items described below. Unrecorded transactions: 1. On May 1,...
-
After several years of losses, Biopure engineers figure out a way to reduce the fixed cost to produce Oxyglobin from $15 million to $10 million. If the price charged to veterinarians is $150 per...
-
In 2016, the price of corn fell and some corn farmers will switch from growing corn in 2017 to growing soybeans. a. Does this fact illustrate the law of demand or the law of supply? Explain your...
-
Suppose that Tommy Hilfigers marginal cost of a jacket is a constant $100 and the total fixed cost at one of its stores is $2,000 a day. This store sells 20 jackets a day, which is its...
-
Leisure Island has 50 hours of labor a day that it can use to produce entertainment and good food. The table shows the maximum quantity of each good that it can produce with different quantities of...
-
Consider a 50-kW rooftop PV system installed at a cost of $3500/kW. The PV system operates with a capacity factor of 22%, $0.005/kWh of maintenance costs, and 1% downtime during a typical year. For a...
-
The price of flavored hake, used by sushi bars in place of crabmeat, has increased by 33 percent over the past three years. Crabmeat is up about 50 percent. Californian sushi rice has increased in...
-
compute the function (x,y) satisfying the eikonal equation d2 + 4y = n and the initial cand b17x (n is a constant parameter). CCX, 1
-
Conduct a VRIO analysis by ranking Husson University (in Maine) business school in terms of the following six dimensions relative to the top three rival schools. If you were the dean with a limited...
-
Propose a mechanism for the basic hydrolysis of benzonitrile to the benzoate ion and ammonia.
-
A student synthesized Compound 1 (below). To purify the compound, he extracted it into aqueous base and then acidified the solution to protonate the acid so that he could extract it back into ether....
-
Suggest the most appropriate method for each of the following laboratory syntheses. In each case, suggest both a chromium reagent and a chromium-free reagent. (a) butan -1-o1 butanal, CH3CH2CH2CHO...
-
Liquefaction can be achieved through (a) Expansion of gas through a work-producing device (isentropic expansion) (b) Joule-Thomson expansion (isenthalpic expansion) (c) Exchange of heat at constant...
-
To estimate the Ozone Depletion Potential of a refrigerant (a) \(\mathrm{CFC}-11\) is used as a reference gas (b) \(\mathrm{CFC}-12\) is used as a reference gas (c) \(\mathrm{CO}_{2}\) is used as a...
-
To compute the Global Warming Potential of a refrigerant (a) HFC-134a is used as a reference gas (b) Hydrocarbon is used as a reference gas (c) \(\mathrm{CO}_{2}\) is used as a reference gas (d)...

Study smarter with the SolutionInn App


