A packed bed absorption tower produces carbonated water for beverages by continuously contacting a stream of...
Fantastic news! We've Found the answer you've been seeking!
Question:

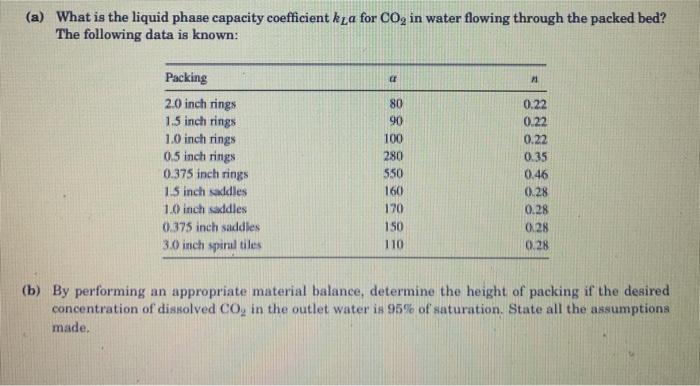
Transcribed Image Text:
A packed bed absorption tower produces carbonated water for beverages by continuously contacting a stream of carbon dioxide (CO₂) gas with a stream of liquid water. Pure mountain spring water, containing no dissolved CO2, enters the top of the column at a flow rate of 5 kmol/min. Pure CO₂ gas at 2 atm is also fed in from the top of the tower at a flow rate of 1 kmol/min. As the water flows down the tower, CO₂ gas absorbs into the water, and the dissolved CO₂ increases down the length of the packed bed. The carbonated water and the unused CO₂ gas exit the bottom of the tower. pure water in packed bed of 1" ceramic rings 1 carbonated water out pure CO, in 2 atm 20°C unused CO, out The tower is packed with 1" ceramic rings and the tower's inner diameter is 0.25 m. The tempera- ture is maintained at 20 C. At this temperature, the Henry's law constant for CO₂ in water is 25.4 -9 m/s, density of liquid water is i atm/(kmol/m), mass diffusivity of dissolved CO, in water is 1.77 * 10 998.2 kg/m³, and dynamic viscosity of water is 9.93×10 kg(ms). 4 (a) What is the liquid phase capacity coefficient ka for CO₂ in water flowing through the packed bed? The following data is known: Packing 2.0 inch rings 1.5 inch rings 1.0 inch rings 0.5 inch rings 0.375 inch rings 1.5 inch saddles 1.0 inch saddles 0.375 inch saddles 3.0 inch spiral tiles a 80 90 100 280 550 160 170 150 110 11 0.22 0.22 0.22 0.35 0.46 0.28 0.28 0.28 0.28 (b) By performing an appropriate material balance, determine the height of packing if the desired concentration of dissolved CO₂ in the outlet water is 95% of saturation. State all the assumptions made. A packed bed absorption tower produces carbonated water for beverages by continuously contacting a stream of carbon dioxide (CO₂) gas with a stream of liquid water. Pure mountain spring water, containing no dissolved CO2, enters the top of the column at a flow rate of 5 kmol/min. Pure CO₂ gas at 2 atm is also fed in from the top of the tower at a flow rate of 1 kmol/min. As the water flows down the tower, CO₂ gas absorbs into the water, and the dissolved CO₂ increases down the length of the packed bed. The carbonated water and the unused CO₂ gas exit the bottom of the tower. pure water in packed bed of 1" ceramic rings 1 carbonated water out pure CO, in 2 atm 20°C unused CO, out The tower is packed with 1" ceramic rings and the tower's inner diameter is 0.25 m. The tempera- ture is maintained at 20 C. At this temperature, the Henry's law constant for CO₂ in water is 25.4 -9 m/s, density of liquid water is i atm/(kmol/m), mass diffusivity of dissolved CO, in water is 1.77 * 10 998.2 kg/m³, and dynamic viscosity of water is 9.93×10 kg(ms). 4 (a) What is the liquid phase capacity coefficient ka for CO₂ in water flowing through the packed bed? The following data is known: Packing 2.0 inch rings 1.5 inch rings 1.0 inch rings 0.5 inch rings 0.375 inch rings 1.5 inch saddles 1.0 inch saddles 0.375 inch saddles 3.0 inch spiral tiles a 80 90 100 280 550 160 170 150 110 11 0.22 0.22 0.22 0.35 0.46 0.28 0.28 0.28 0.28 (b) By performing an appropriate material balance, determine the height of packing if the desired concentration of dissolved CO₂ in the outlet water is 95% of saturation. State all the assumptions made.
Expert Answer:
Answer rating: 100% (QA)
a Liquid Phase Capacity Coefficient kLa Calculation The liquid phase capacity coefficient kLa is a measure of the mass transfer efficiency in the absorption process It is given by the equation kLa 2 o... View the full answer

Related Book For 

Fundamentals Of Momentum Heat And Mass Transfer
ISBN: 9781119723547
7th Edition
Authors: James Welty, Gregory L. Rorrer, David G. Foster
Posted Date:
Students also viewed these physics questions
-
A process is being developed to produce carbonated beverages. As part of this process, a packed-bed absorption tower will be used to dissolve carbon dioxide, CO 2 , gas into water. Pure mountain...
-
A process is being developed to produce carbonated beverages. As part of this process, a wetted-wall absorption column 2.0 m long will be used to dissolve carbon dioxide (CO 2 ) gas into water. The...
-
Beaver Brewing Corporation has received a contract to produce carbonated beverages. A packed-bed absorption tower will be used for dissolving carbon dioxide (CO 2 ) gas into water. In the present...
-
Repeat Prob. 37 using singularity functions exclusively (including reactions). Repeat Prob. 37, For the beam shown, find the reactions at the supports and plot the shear-force and bending-moment...
-
Albert Machine Tool Company produces tools on a job order basis. During May, two jobs were completed, and the following costs were incurred: Other factory costs for the month totaled $16,800. Factory...
-
We are designing a new process that uses natural gas, which will be delivered via pipeline at conditions of P = 1000 psia and T = 100F. In order to size process equipment, we need to know the density...
-
Reconsider the data from Problem 56. What is the capital recovery cost of Model 334A? Data from problem 56 Octavia Bakery is planning to purchase one of two ovens. The expected cash flows for each...
-
During the second half of December 20-1, TJ's Specialty Shop engaged in the following transactions: Dec. 16 Received payment from Lucy Greene on account, $1,960. 16 Sold merchandise on account to Kim...
-
Write a C program that asks the user to enter the sum and difference of 2 integer numbers. The program will find the 2 numbers and display them as shown below. Input validation: sum plus difference...
-
How do you calculate the percentage of change in sales from 20X8 to 20X9? This will go in cell D7? Show the formula. Kretsmart Common-Size Income Statements For the Years Ended December 31, 20'X'9,...
-
The current in the 8.00-2 resistor in the drawing is 0.645 A. Find the current in (a) the 20.0-2 resistor and in (b) the 9.00-2 resistor. 8.00 a ww- 20.02 16.0 a 9.00 2 18.0 2
-
Network models such as PERT and CPM are used a. to plan large and complex projects. b. to schedule large and complex projects. c. to monitor large and complex projects. d. to control large and...
-
Simulation is a technique to imitate real-world situations with mathematical models not affecting operations. a. True b. False
-
In analyzing a problem, you should normally study a. the qualitative aspects. b. the quantitative aspects. c. both a and b. d. neither a nor b.
-
Early European airlines carried passengers. Why werent passengers carried on the first U.S. commercial flights?
-
Using simulation for a queuing problem would be appropriate if a. the arrival rate follows a Poisson distribution. b. the service rate is constant. c. the FIFO queue discipline is assumed. d. there...
-
How does a for loop start? OA. for (i=0; i
-
A non-charmed baryon has strangeness S = 2 and electric charge Q = 0. What are the possible values of its isospin I and of its third component I z ? What is it usually called if I = 1/2?
-
Air rapidly flows over a 10.0 m x 8.0 m rectangular spill of gasoline (n-octane) at a velocity of 8.0 m/s. The spill occurred within an enclosure where the gas temperature was maintained at 27 C. You...
-
Consider the cylindrical porous pellet shown in the figure below. A gas mixture of species A and B continually flows over the top face of the pellet. The sides and bottom of the pellet are sealed,...
-
The soil venting shown in the figure (next page) is used to treat soil contaminated with volatile, toxic liquids. In the present situation, the porous soil particles are saturated with liquid TCE, a...
-
Suppose you want to create a price index for the price of a personal pizza across several Canadian university campuses, as of March 1 , 201 6. The data are as follows: a. Using Calgary as the "base...
-
Fill in the blanks to make the following statements correct. a. Ceteris paribus, the price of a product and the quantity demanded are related ________ b. Ceteris paribus, the price of a product and...
-
The following questions will provide practice working with simple linear functions. All questions refer to a coordinate graph with the variable \(X\) on the horizontal axis and the variable \(Y\) on...

Study smarter with the SolutionInn App


