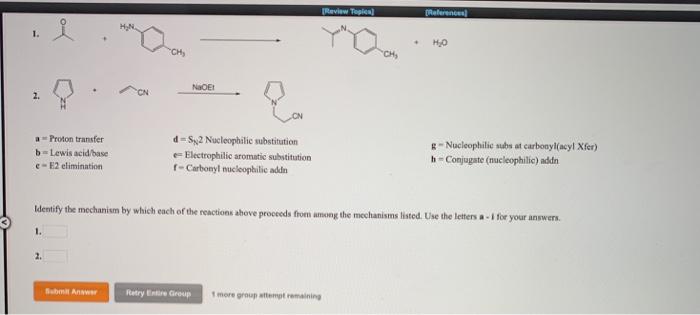
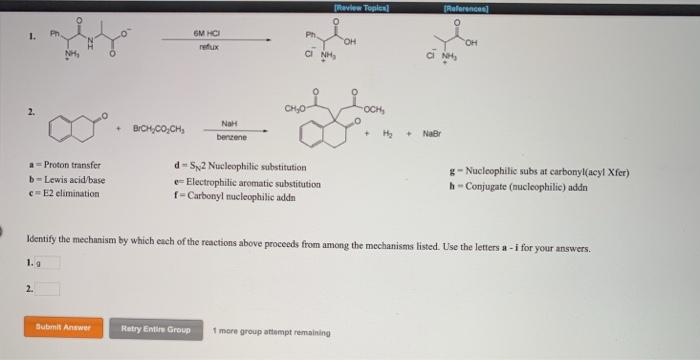
Question: a Proton transfer b-Lewis acid/base e-E2 elimination 1. 2. CH Submit Answer NOT d-S2 Nucleophilic substitution - Electrophilic aromatic substitution f-Carbonyl nucleophilic addn [Review
![Topic] "CH Retry Entire Group 1 more group attempt remaining * Identify](https://dsd5zvtm8ll6.cloudfront.net/si.experts.images/2023/02/63ddf3280bb7d_1675490084881.jpg)
a Proton transfer b-Lewis acid/base e-E2 elimination 1. 2. CH Submit Answer NOT d-S2 Nucleophilic substitution - Electrophilic aromatic substitution f-Carbonyl nucleophilic addn [Review Topic] "CH Retry Entire Group 1 more group attempt remaining * Identify the mechanism by which each of the reactions above proceeds from among the mechanisms listed. Use the letters a-1 for your answers. [References] HO g-Nucleophilic subs at carbonyl(acy! Xfer) h-Conjugate (nucleophilic) addn lifo a Proton transfer b-Lewis acid/base c=E2 elimination 2. BICH,COCH Submit Answer 6M HCI refux NaH benzene Retry Entire Group Ph d-S2 Nucleophilic substitution e-Electrophilic aromatic substitution f-Carbonyl mucleophilic addn CINH [Review Topics] 1 more group attempt remaining OH + H [References] ci Not NaBr Identify the mechanism by which each of the reactions above proceeds from among the mechanisms listed. Use the letters a-i for your answers. 1.9 OH g-Nucleophilic subs at carbonyl(acyl Xfer) h-Conjugate (nucleophilic) addn 2. OH 1. a = Proton transfer b-Lewis acid/base c-E2 elimination 2. OH Submit Answer NH Biosynthesis of adreneline (epinephrine) OCCHCHCH- NH OH NH d=SN2 Nucleophilic substitution e- Electrophilic aromatic substitution f-Carbonyl nucleophilic addn PROVIEW Topic Retry Entire Group 1 more group attempt remaining References HO Identify the mechanism by which each of the reactions above proceeds from among the mechanisms listed. Use the letters a-i for your answers. NHCH, 0,CCH.CH,CH, S HOH H g=Nucleophilic subs at carbonyl(acyl Xfer) h-Conjugate (nucleophilic) addn H NH
Step by Step Solution
3.41 Rating (157 Votes )
There are 3 Steps involved in it
The detailed ... View full answer

Get step-by-step solutions from verified subject matter experts


