A ramjet engine operates as follows: 1-2: air is decelerated isentropically in a diffuser 2-3: fuel...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
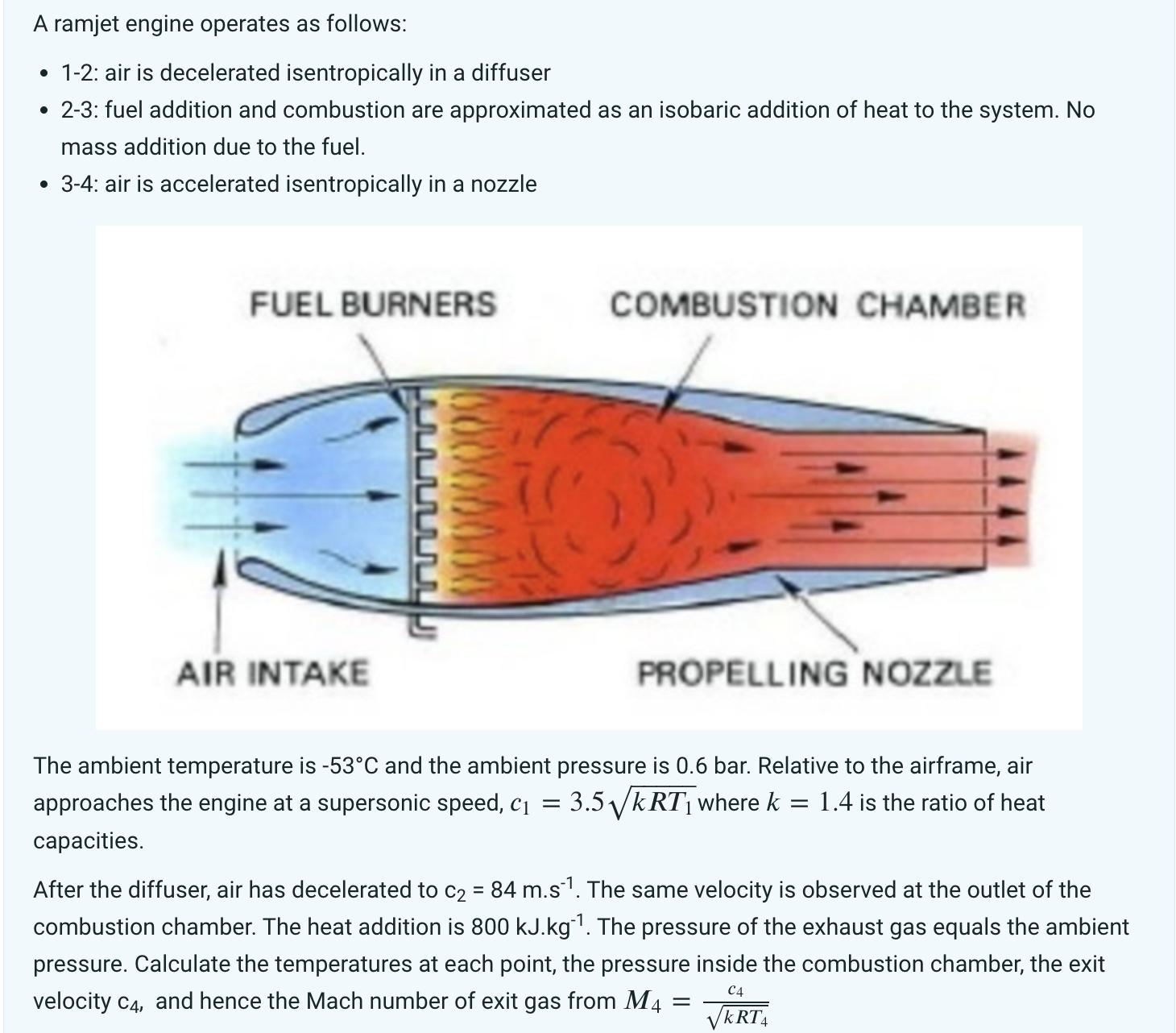
A ramjet engine operates as follows: 1-2: air is decelerated isentropically in a diffuser 2-3: fuel addition and combustion are approximated as an isobaric addition of heat to the system. No mass addition due to the fuel. 3-4: air is accelerated isentropically in a nozzle FUEL BURNERS AIR INTAKE COMBUSTION CHAMBER PROPELLING NOZZLE The ambient temperature is -53C and the ambient pressure is 0.6 bar. Relative to the airframe, air approaches the engine at a supersonic speed, C = 3.5KRT1 where k = 1.4 is the ratio of heat capacities. After the diffuser, air has decelerated to c = 84 m.s1. The same velocity is observed at the outlet of the combustion chamber. The heat addition is 800 kJ.kg 1. The pressure of the exhaust gas equals the ambient pressure. Calculate the temperatures at each point, the pressure inside the combustion chamber, the exit velocity C4, and hence the Mach number of exit gas from M4 C4 = KRT4 Maximum marks will be awarded as follows: a) A succinct problem statement (1 mark) b) A plant diagram (3 marks) c) A T-S diagram (3 marks) d) A list of assumptions made to solve the question (2 marks) e) A list of physical laws used to solve the question (2 marks) f) Calculations (17 marks) 1. Calculate T2, T3 and T4. (9 marks) 2. Calculate the pressure in the combustion chamber. (3 marks) 3. Calculate the exit velocity. (3 marks) 4. Calculate the exit Mach number. (2 marks) A ramjet engine operates as follows: 1-2: air is decelerated isentropically in a diffuser 2-3: fuel addition and combustion are approximated as an isobaric addition of heat to the system. No mass addition due to the fuel. 3-4: air is accelerated isentropically in a nozzle FUEL BURNERS AIR INTAKE COMBUSTION CHAMBER PROPELLING NOZZLE The ambient temperature is -53C and the ambient pressure is 0.6 bar. Relative to the airframe, air approaches the engine at a supersonic speed, C = 3.5KRT1 where k = 1.4 is the ratio of heat capacities. After the diffuser, air has decelerated to c = 84 m.s1. The same velocity is observed at the outlet of the combustion chamber. The heat addition is 800 kJ.kg 1. The pressure of the exhaust gas equals the ambient pressure. Calculate the temperatures at each point, the pressure inside the combustion chamber, the exit velocity C4, and hence the Mach number of exit gas from M4 C4 = KRT4 Maximum marks will be awarded as follows: a) A succinct problem statement (1 mark) b) A plant diagram (3 marks) c) A T-S diagram (3 marks) d) A list of assumptions made to solve the question (2 marks) e) A list of physical laws used to solve the question (2 marks) f) Calculations (17 marks) 1. Calculate T2, T3 and T4. (9 marks) 2. Calculate the pressure in the combustion chamber. (3 marks) 3. Calculate the exit velocity. (3 marks) 4. Calculate the exit Mach number. (2 marks)
Expert Answer:

Related Book For 

Thermodynamics An Interactive Approach
ISBN: 978-0130351173
1st edition
Authors: Subrata Bhattacharjee
Posted Date:
Students also viewed these chemical engineering questions
-
How do markets respond to price ceilings and price floors? Do attempts to repeal the laws of supply and demand meet their objectives? 52. How does the United States differ from the European Union in...
-
A particle is described by the normalized wave function (x, y, z) = Axe-ax2 e -y2 e-yz2', where A, a, , and are all real, positive constants. The probability that the particle will be found in the...
-
Klein Corporation manufactures two models of watches. Model Wonder displays cartoon characters and has simple features designed for kids. Model Marvel has sophisticated features such as dual time...
-
(a) What was the price difference between U.S. and Chinese solar panels in 2013? (b) How much money did U.S. consumers save as a result of this price difference if they purchased 2,000 megawatts of...
-
When the Concepcions went to AT&T to get a phone, they were confused as to why they were paying money for the phone when it was advertised as being free. They had to pay sales tax on the full normal...
-
The partners in New Yorker Company decide to liquidate the firm when the balance sheet shows the following The partners share income and loss 5 : 3 : 2. During the process of liquidation, the...
-
The Oracle Database Upgrade Assistant (DBUA) is a GUI tool but can also run in silent command line mode. As a DBA, in what circumstances would you recommend using silent mode?
-
Write a paper about the graphic designer david carson.
-
What is the difference between a single payer system and a private firm ? Also, according to the video, health care systems are evaluated by three criteria - what are they? What are some of the...
-
Two companies are competing in the market. There are no other companies in this market in this field. If company A has reasonable information about how much company B will produce. As well as that A...
-
1. Print the number of fruits in each category 2. Print the percent of the total that each fruit Market = [1 Apple','2 Apple','3 Apple','4 Orange','5 Banana','6 Orange','7 Orange','8 Apple','9...
-
At an amusement park, 66 of 90 rides have a height restriction. What fraction of the rides, in its simplest form, has a height restriction? 2. A bicycle rider is riding at a rate of 15 km/h for...
-
The general objective of the assignment is the analysis of a case study at the students' choice related to the Knowledge Management system in a company/organization. Students are required to focus...
-
Consider a book shelf of width, w, and length, L. It is desired to minimize the mass of the shelf while maintaining the maximum deflection,, below a critical value of 12L3 CEwt properties should be...
-
Ball bearings are widely used in industrial applications. You work for an industrial food machinery manufacturer and your role is to design the driveshaft assembly on a new type of equipment that...
-
Steam at 400oC enters a nozzle with an average velocity of 20 m/s. If the specific volume (v) and the flow area at the inlet are measured as 0.1 m2/kg and 0.01 m2 respectively, determine (a) The...
-
Nitrogen gas at 400 K and 300 kPa behaves as an ideal gas. Estimate the cp and cv of nitrogen at this state.
-
A system contains an unknown solid of unknown mass, which is heated from 300 K to 900 K at 100 kPa. The change in internal energy (U) of the system is measured as 61.02 MJ. Using the SL model,...
-
Should morality, in and of itself, be considered a sufficient basis for defining particular conduct as criminal?
-
What are the chief distinctions between the civil and criminal law? Why do the criminal and civil law sometimes overlap?
-
What means of punishment for criminal offenses exist in your state? Is capital punishment available for persons convicted of first-degree murder? Which punishments, if any, do you think are most...

Study smarter with the SolutionInn App


