A. Rank the following compounds in order of decreasingacidity, putting the most acidic first. why? CH 3
Question:
A. Rank the following compounds in order of decreasingacidity, putting the most acidic first. why?
CH3OCH3 CH3CHO CH3CH2OH CH3COOH
I II III IV
B. Which of the following matches the molecules to thenames of the functional groups? why?
CH3OCH3 Ether
CH3CONH2 Amine
CH3SH Thiol
CH3CHO Alcohol
C. Which of the following compounds has the highestboiling point? why?
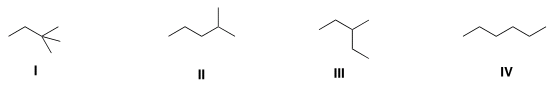
D. Which of the following molecules are chiral?why?
I. 2-Chlorobutane
II. 3-Bromopentane
III. 2-Methylpropene
IV. 2-Bromo-3-methylbutane
E. What is the relationship between the following 2components?? why?
![]()
| Constitutional isomers |
| Identical |
| Stereoisomers |
| Not isomers, different compounds |
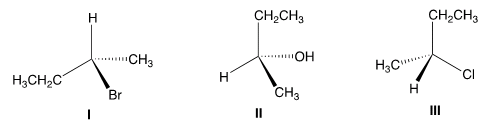
F. Which of the following has the S Configuration?Why?
G. What kind of reaction does theconversion of A to B represent?
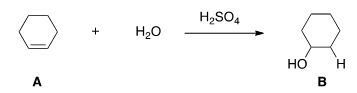
| Acid-base reaction. |
| Elimination reaction. |
| Substitution reaction. |
| Addition reaction. |
H. For the following reaction, usethe identity of the alkyl halide and nucleophile to determine whichsubstitution mechanism occurs. Then determine which solvent affordsthe faster reaction.
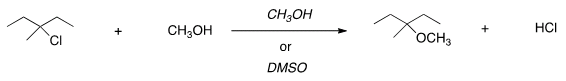
| SN1, CH3OH |
| SN1, DMSO |
| SN2, CH3OH |
| SN2, DMSO |
I. Which of the following is most likely to reactas a nucleophile rather a base?
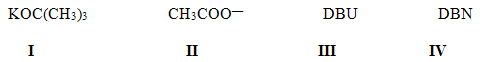
J. What is the most likely mechanism for thereaction below?
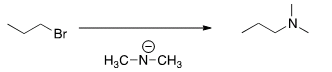
| SN1 |
| SN2 |
| E1 |
| E2 |
What is the best choice of reagent to accomplish thefollowing transformation?

| HCl |
| SOCl2, pyridine |
| Cl2 |
| H3O+, H2O |





