Using the data in the ideal gas tables, determine the equilibrium constant Kp for the reaction:...
Fantastic news! We've Found the answer you've been seeking!
Question:

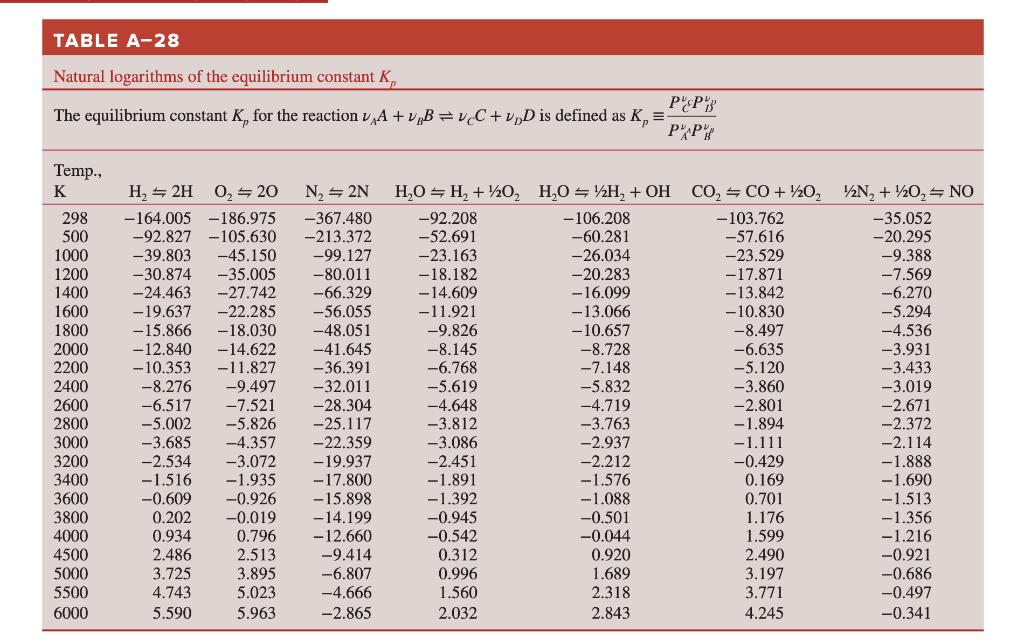
Transcribed Image Text:
Using the data in the ideal gas tables, determine the equilibrium constant Kp for the reaction: H₂O = 1/2 H₂ + OH at (a) 298 K and (b) 3000 K. Compare your results with the Kp values listed in Table A-28. TABLE A-28 Natural logarithms of the equilibrium constant K PEP PAP The equilibrium constant K, for the reaction A + B =vC+v₂D is defined as K₂ = Temp., K 298 500 1000 1200 1400 1600 1800 2000 2200 2400 2600 2800 3000 3200 3400 3600 3800 4000 4500 5000 5500 6000 H₂ → 2H 0₂ 20 N₂ = 2N -367.480 -164.005 -186.975 -92.827 -105.630 -213.372 -99.127 -39.803 -45.150 -30.874 -35.005 -80.011 -24.463 -27.742 -66.329 -19.637 -22.285 -56.055 -15.866 -18.030 -48.051 -12.840 -14.622 -41.645 -10.353 -11.827 -36.391 -8.276 -9.497 -32.011 -6.517 -7.521 -28.304 -5.002 -5.826 -25.117 -3.685 -4.357 -22.359 -2.534 -3.072 -19.937 -1.516 -1.935 -17.800 -0.609 -0.926 -15.898 0.202 -0.019 -14.199 0.934 0.796 -12.660 2.486 2.513 -9.414 3.725 3.895 -6.807 4.743 5.023 -4.666 5.590 5.963 -2.865 H₂O H₂ + 1/2O₂ H₂O = ½H₂ + OH CO₂=CO+ //20₂2 2N₂ + 1/2O₂ = NO -92.208 -103.762 -35.052 -52.691 -57.616 -20.295 -23.163 -23.529 -9.388 -18.182 -17.871 -7.569 -14.609 -13.842 -6.270 -11.921 -10.830 -5.294 -9.826 -8.497 -4.536 -8.145. -6.635 -3.931 -6.768 -5.120 -3.433 -5.619 -3.860 -3.019 -4.648 -2.801 -2.671 -3.812 -1.894 -2.372 -3.086 -1.111 -2.114 -2.451 -0.429 -1.888 -1.891 0.169 -1.690 -1.392 0.701 -0.945 1.176 -0.542 1.599 0.312 2.490 0.996 3.197 1.560 3.771 2.032 4.245 -106.208 -60.281 -26.034 -20.283 - 16.099 -13.066 -10.657 -8.728 -7.148 -5.832 -4.719 -3.763 -2.937 -2.212 -1.576 -1.088 -0.501 -0.044 0.920 1.689 2.318 2.843 -1.513 -1.356 -1.216 -0.921 -0.686 -0.497 -0.341 Using the data in the ideal gas tables, determine the equilibrium constant Kp for the reaction: H₂O = 1/2 H₂ + OH at (a) 298 K and (b) 3000 K. Compare your results with the Kp values listed in Table A-28. TABLE A-28 Natural logarithms of the equilibrium constant K PEP PAP The equilibrium constant K, for the reaction A + B =vC+v₂D is defined as K₂ = Temp., K 298 500 1000 1200 1400 1600 1800 2000 2200 2400 2600 2800 3000 3200 3400 3600 3800 4000 4500 5000 5500 6000 H₂ → 2H 0₂ 20 N₂ = 2N -367.480 -164.005 -186.975 -92.827 -105.630 -213.372 -99.127 -39.803 -45.150 -30.874 -35.005 -80.011 -24.463 -27.742 -66.329 -19.637 -22.285 -56.055 -15.866 -18.030 -48.051 -12.840 -14.622 -41.645 -10.353 -11.827 -36.391 -8.276 -9.497 -32.011 -6.517 -7.521 -28.304 -5.002 -5.826 -25.117 -3.685 -4.357 -22.359 -2.534 -3.072 -19.937 -1.516 -1.935 -17.800 -0.609 -0.926 -15.898 0.202 -0.019 -14.199 0.934 0.796 -12.660 2.486 2.513 -9.414 3.725 3.895 -6.807 4.743 5.023 -4.666 5.590 5.963 -2.865 H₂O H₂ + 1/2O₂ H₂O = ½H₂ + OH CO₂=CO+ //20₂2 2N₂ + 1/2O₂ = NO -92.208 -103.762 -35.052 -52.691 -57.616 -20.295 -23.163 -23.529 -9.388 -18.182 -17.871 -7.569 -14.609 -13.842 -6.270 -11.921 -10.830 -5.294 -9.826 -8.497 -4.536 -8.145. -6.635 -3.931 -6.768 -5.120 -3.433 -5.619 -3.860 -3.019 -4.648 -2.801 -2.671 -3.812 -1.894 -2.372 -3.086 -1.111 -2.114 -2.451 -0.429 -1.888 -1.891 0.169 -1.690 -1.392 0.701 -0.945 1.176 -0.542 1.599 0.312 2.490 0.996 3.197 1.560 3.771 2.032 4.245 -106.208 -60.281 -26.034 -20.283 - 16.099 -13.066 -10.657 -8.728 -7.148 -5.832 -4.719 -3.763 -2.937 -2.212 -1.576 -1.088 -0.501 -0.044 0.920 1.689 2.318 2.843 -1.513 -1.356 -1.216 -0.921 -0.686 -0.497 -0.341
Expert Answer:
Answer rating: 100% (QA)
Write the reactions at 5000 K as follows HO H OH K 1689 1 1 HO H 0 K 0... View the full answer

Related Book For 

An Introduction to Derivative Securities Financial Markets and Risk Management
ISBN: 978-0393913071
1st edition
Authors: Robert A. Jarrow, Arkadev Chatterjee
Posted Date:
Students also viewed these mechanical engineering questions
-
Elfalan Corporation produces a single product. The cost of producing and selling a single unit of this product at the company's normal activity level of 44,000 units per month is as follows: Direct...
-
The Sweeney Paper Company is planning to sell $10 million worth of long-term bonds with an 11% interest rate. The company believes that it can sell the $ 1,000 par value bonds at a price that will...
-
A manufacturing company is planning to sell 1,200 boxes of ceramic tile, with production estimated at 1,120 boxes during May. Each box of tile requires 44 pounds of clay mix and a quarter hour of...
-
A 1-m3 insulated tank contains air at 1 MPa, 560 K. The tank is now discharged through a small convergent nozzle to the atmosphere at 100 kPa. The nozzle has an exit area of2 x 10-5 m2. a. Find the...
-
Is the gift you purchased for that special someone really appreciated? This was the question of interest to business professors at Stanford University. Their research was published in the Journal of...
-
Your boss is considering the use of a new predictor. The base rate is high, the selection ratio is low, and the validity coefficient is high for the current predictor. What would you advise your boss...
-
The accountant for Flenderson Roofing, Inc., posted adjusting entries (a) through (e) to the accounts at December 31, 2010. Selected balance sheet accounts and all the revenues and expenses of the...
-
BPO Services is in the business of digitizing information from forms that are filled out by hand. In 2006, a big client gave BPO a distribution of the forms that it digitized in house last year, and...
-
The domain of f(x) is {XER) and the domain of f^' (x) is {XERIX=2} sketch what f(x) can be and justify why.
-
Problem Statement: The aim of this case study is to propose a daily capacity level and level of resourcing required for Q2 & Q3 2023. The proposed daily capacity level should achieve the highest...
-
Using a car as a model of a rigid body, briefly describe with a diagram the terms of a rigid body used in the study of statics for a two-dimensional equilibrium condition. Show all relevant forces...
-
The very long cylindrical solenoid of Figure P29.27 has a radius of \(0.50 \mathrm{~m}\) and 1200 windings per meter along its length. A circular conducting loop of radius \(1.0 \mathrm{~m}\), and...
-
Which of the following is the repeatability of scientific observations? a. Reliability b. Validity c. Theory d. Integrity
-
Under common law, a CPA may be liable to a client. a. Explain the meaning and importance of the term privity of contract. b. How may an auditor breach a contract? c. What cause ordinarily underlies a...
-
Which data-collection method gathers data from a relatively large number of cases at a particular time? a. Survey b. Observation c. Essay d. Experimental study
-
a. What were the critical issues in (a) the Contine, ital Vending case and (b ) the BarChris case? b. What action was taken by the profession as a result of each case?
-
On the off chance that you are a financial backer, high-recurrence exchanging (HFT) is a piece of your life regardless of whether you know it. You've probably bought shares presented by a PC, or sold...
-
Review Exhibit 11.4. Analyze each product on the graph according to the characteristics that influence the rate of adoption. For example, what can you conclude from the data about the relative...
-
What is an efficient market, and what does it mean for a market to be weak-form, semi strong-form, and strong-form efficient?
-
Suppose that Jewelry Company is planning to sell twenty thousand ounces of platinum at some future date. The standard deviation of changes in the futures price per ounce sdF is 12.86, that for...
-
Compute the weekly 95 percent and 99 percent value- at- risk for a portfolio consisting of the three stocks in equal proportions.
-
Paying an account payable will a. Increase one asset and decrease another asset b. Decrease an asset and decrease owner's equity c. Decrease an asset and decrease a liability d. Increase an asset and...
-
The financial statement that summarizes assets, liabil ties, and owner's equity is called the a. Cash flow statement b. Balance sheet c. Income statement d. Statement of owner's equity
-
The financial statements that are dated for a time perio (rather than for a specific point in time) are the a. Balance sheet and income statement b. Balance sheet and statement of owner's equity c....

Study smarter with the SolutionInn App


